|
A.S.S.E.Psi.
web site (History of Psychiatry and Psychoanalytic Psychotherapy
)
A.S.S.E.Psi.NEWS
(to subscribe our monthly newsletter)
Ce.Psi.Di. (Centro
di Psicoterapia Dinamica "Mauro Mancia")
Maitres
� dispenser (Our reviews about psychoanalytic congresses)
Biblio
Reviews (Recensioni)
Congressi
ECM (in italian)
Events
(our congresses)
Tatiana Rosenthal
and ... other 'psycho-suiciders'
Thalassa.
Portolano of Psychoanalysis
PsychoWitz - Psychoanalysis and Humor (...per ridere un po'!)
Giuseppe Leo's Art
Gallery
Spazio
Rosenthal (femininity and psychoanalysis)
Psicoanalisi
Europea Video
Channel
A.S.S.E.Psi. Video
Channel
Ultima uscita/New issue:
"PSYCHANALYSE,
LIEUX DE M�MOIRE ET TRAUMATISMES COLLECTIFS"

�crits de:
J.
Altounian, S. Amati Sas, M. Avakian, W. Bohleber, M. El Husseini,
R. El Khayat, Y. Gampel, R. Ka�s, J. Kristeva, G. Leo, A.
Loncan,
P. Matvejević, M.-R. Moro
Maison d'�dition: Edizioni Frenis Zero
ISBN: 978-88-97479-29-1
Anno/Year: 2020
Pages: 482
Prezzo/Price: � 40,00
Achater sur
Amazon.fr
"FEAR
OF LOCKDOWN. PSYCHOANALYSIS, PANDEMIC DISCONTENTS AND CLIMATE
CHANGE"

Writings by: H.
Catz, A. Ferruta, M. Francesconi, P. R. Goisis,
G. Leo, N. McWilliams, G. Riefolo, M.
Roth, C. Schinaia, D. Scotto di Fasano
Editore/Publisher: Edizioni Frenis Zero
ISBN: 978-88-97479-21-5
Anno/Year: 2020
Pages: 296
Prezzo/Price: � 45,00
Click
here to have a preview
Click
here to order the book/Ordina
su Amazon.it
"Psicoanalisi e luoghi della memoria" a cura di Giuseppe Leo
(editor)

Writings by: J.
Altounian, S. Amati Sas, M. Avakian, W. Bohleber, M. Breccia, A.
Coen, A. Cusin, G. Dana, J. Deutsch, S. Fizzarotti Selvaggi, Y.
Gampel, H. Halberstadt-Freud, N. Janigro, R. Ka�s, G. Leo, M.
Maisetti, F. Mazzei, M. Ritter, C. Trono, S. Varvin e H.-J. Wirth
Editore/Publisher: Edizioni Frenis Zero
ISBN: 978-88-903710-2-8
Anno/Year: 2020 (2 ed.)
Pages: 545
Prezzo/Price: � 41,00
Click
here to have a preview
Click
here to order the book/Ordina
su Amazon.it


Rock
Music & Psychoanalysis
Edited
by: Giuseppe Leo
Authored
by/autori: Lewis Aron Heather
Ferguson Joseph LeDoux Giuseppe Leo Rod Tweedy
Editore/Publisher:
Edizioni Frenis Zero
Collection/Collana: Borders
of Psychoanalysis
Anno/Year:
2020
Pagine/Pages:
201
ISBN:978-88-97479-20-8
Click
here to order the book

"Essere
nella cura"
Authored
by/autori: Giacomo Di Marco & Isabella
Schiappadori
Editore/Publisher:
Edizioni Frenis Zero
Collection/Collana: Confini
della Psicoanalisi
Anno/Year:
2019
Pagine/Pages:
210
ISBN:978-88-97479-17-8
Click
here to order the book

"Enactment
in Psychoanalysis"
Edited
by Giuseppe Leo & Giuseppe Riefolo
Writings
by: E. Ginot J.R.
Greenberg J. Kraus J.D. Safran
Publisher:
Frenis Zero
Collection:
Borders of Psychoanalysis
Year:
2019
Pages:
326
ISBN: 978-88-97479-15-4
Click
here to order the book

"Infant
Research and Psychoanalysis"
Edited
by Giuseppe Leo
Writings
by: B. Beebe K.
Lyons-Ruth J. P. Nahum E. Solheim C.
Trevarthen E. Z. Tronick L.
Vulliez-Coady
Publisher:
Frenis Zero
Collection:
Borders of Psychoanalysis
Year:
2018
Pages:
273
ISBN: 978-88-97479-14-7
Click
here to order the book

"Fundamentalism
and Psychoanalysis"
Edited
by Giuseppe Leo
Prefaced
by: Vamik D. Volkan
Writings
by: L. Auestad W.
Bohleber S. Varvin L. West
Publisher:
Frenis Zero
Collection:
Mediterranean Id-entities
Year:
2017
Pages:
214
ISBN: 978-88-97479-13-0
Click
here to order the book

"Psicoanalisi, luoghi della resilienza
ed immigrazione"
Edited
by/a cura di:
Giuseppe Leo
Writings by/scritti di:
S.
Ara�jo Cabral, L.
Curone,
M. Francesconi,
L.
Frattini,
S.
Impagliazzo,
D. Centenaro Levandowski, G. Magnani, M. Manetti, C. Marangio,
G. A. Marra e Rosa, M. Martelli, M. R. Moro,
R. K. Papadopoulos, A. Pellicciari,
G. Rigon,
D.
Scotto di Fasano,
E. Zini, A. Zunino
Editore/Publisher: Edizioni Frenis Zero
Collection/Collana: Mediterranean
Id-entities
Anno/Year:
2017
Pagine/Pages:
372
ISBN:978-88-97479-11-6

"Psicoanalisi in Terra Santa"
Edited
by/a cura di: Ambra Cusin & Giuseppe Leo
Prefaced by/prefazione
di:
Anna Sabatini Scalmati
Writings by/scritti di:
H. Abramovitch A. Cusin M. Dwairy A. Lotem M.
Mansur M. P. Salatiello Afterword
by/ Postfazione
di:
Ch. U. Schminck-Gustavus
Notes by/ Note di: Nader Akkad
Editore/Publisher: Edizioni Frenis Zero
Collection/Collana: Mediterranean
Id-entities
Anno/Year:
2017
Pagine/Pages:
170
ISBN:978-88-97479-12-3

"Essere bambini a Gaza. Il trauma
infinito"
Authored
by/autore: Maria Patrizia Salatiello
Editore/Publisher: Edizioni Frenis Zero
Collection/Collana: Mediterranean
Id-entities
Anno/Year:
2016
Pagine/Pages:
242
ISBN:978-88-97479-08-6

Psychoanalysis,
Collective Traumas and Memory Places (English Edition)
Edited
by/a cura di: Giuseppe Leo Prefaced by/prefazione
di:
R.D.Hinshelwood
Writings by/scritti di: J. Altounian
W. Bohleber J. Deutsch
H. Halberstadt-Freud Y. Gampel
N. Janigro R.K. Papadopoulos
M. Ritter S. Varvin H.-J. Wirth
Editore/Publisher: Edizioni Frenis Zero
Collection/Collana: Mediterranean
Id-entities
Anno/Year:
2015
Pagine/Pages:
330
ISBN:978-88-97479-09-3

"L'uomo
dietro al lettino" di
Gabriele Cassullo
Prefaced
by/prefazione di: Jeremy
Holmes
Editore/Publisher: Edizioni Frenis Zero
Collection/Collana: Biografie
dell'Inconscio
Anno/Year:
2015
Pagine/Pages:
350
ISBN:978-88-97479-07-9
Prezzo/Price:
� 29,00
Click
here to order the book
(per Edizione
rilegata- Hardcover clicca qui)

"Neuroscience
and Psychoanalysis" (English Edition)
Edited by/a cura di: Giuseppe Leo Prefaced by/prefazione
di: Georg Northoff
Writings by/scritti di: D. Mann
A. N. Schore R. Stickgold
B.A. Van Der Kolk G. Vaslamatzis M.P. Walker
Editore/Publisher: Edizioni Frenis Zero
Collection/Collana: Psicoanalisi e neuroscienze
Anno/Year: 2014
Pagine/Pages: 300
ISBN:978-88-97479-06-2
Prezzo/Price: � 49,00
Click
here to order the book

Vera
Schmidt, "Scritti su psicoanalisi infantile ed
educazione"
Edited by/a cura di: Giuseppe Leo Prefaced by/prefazione
di: Alberto Angelini
Introduced by/introduzione di: Vlasta Polojaz
Afterword by/post-fazione di: Rita Corsa
Editore/Publisher: Edizioni Frenis Zero
Collana: Biografie dell'Inconscio
Anno/Year: 2014
Pagine/Pages: 248
ISBN:978-88-97479-05-5
Prezzo/Price: � 29,00
Click
here to order the book

Resnik,
S. et al. (a cura di Monica Ferri), "L'ascolto dei
sensi e dei luoghi nella relazione terapeutica"
Writings by:A.
Ambrosini, A. Bimbi, M. Ferri, G.
Gabbriellini, A. Luperini, S. Resnik,
S. Rodighiero, R. Tancredi, A. Taquini Resnik,
G. Trippi
Editore/Publisher: Edizioni Frenis Zero
Collana: Confini della Psicoanalisi
Anno/Year: 2013
Pagine/Pages: 156
ISBN:978-88-97479-04-8
Prezzo/Price: � 37,00
Click
here to order the book
Silvio
G. Cusin, "Sessualit� e conoscenza"

A cura di/Edited by: A. Cusin & G. Leo
Editore/Publisher: Edizioni Frenis Zero
Collana/Collection: Biografie dell'Inconscio
Anno/Year: 2013
Pagine/Pages: 476
ISBN: 978-88-97479-03-1
Prezzo/Price:
� 39,00
Click
here to order the book
AA.VV.,
"Psicoanalisi e luoghi della riabilitazione", a cura
di G. Leo e G. Riefolo (Editors)

A cura di/Edited by: G. Leo & G. Riefolo
Editore/Publisher: Edizioni Frenis Zero
Collana/Collection: Id-entit� mediterranee
Anno/Year: 2013
Pagine/Pages: 426
ISBN: 978-88-903710-9-7
Prezzo/Price:
� 39,00
Click
here to order the book
AA.VV.,
"Scrittura e memoria", a cura di R. Bolletti (Editor)

Writings by: J.
Altounian, S. Amati Sas, A. Arslan, R. Bolletti, P. De
Silvestris, M. Morello, A. Sabatini Scalmati.
Editore/Publisher: Edizioni Frenis Zero
Collana: Cordoglio e pregiudizio
Anno/Year: 2012
Pagine/Pages: 136
ISBN: 978-88-903710-7-3
Prezzo/Price: � 23,00
Click
here to order the book
AA.VV., "Lo
spazio velato. Femminile e discorso
psicoanalitico"
a cura di G. Leo e L. Montani (Editors)

Writings by: A.
Cusin, J. Kristeva, A. Loncan, S. Marino, B.
Massimilla, L. Montani, A. Nunziante Cesaro, S.
Parrello, M. Sommantico, G. Stanziano, L.
Tarantini, A. Zurolo.
Editore/Publisher: Edizioni Frenis Zero
Collana: Confini della psicoanalisi
Anno/Year: 2012
Pagine/Pages: 382
ISBN: 978-88-903710-6-6
Prezzo/Price: � 39,00
Click
here to order the book
AA.VV., Psychoanalysis
and its Borders, a cura di
G. Leo (Editor)

Writings by: J. Altounian, P.
Fonagy, G.O. Gabbard, J.S. Grotstein, R.D. Hinshelwood, J.P.
Jimenez, O.F. Kernberg, S. Resnik.
Editore/Publisher: Edizioni Frenis Zero
Collana/Collection: Borders of Psychoanalysis
Anno/Year: 2012
Pagine/Pages: 348
ISBN: 978-88-974790-2-4
Prezzo/Price: � 19,00
Click
here to order the book
AA.VV.,
"Psicoanalisi e luoghi della negazione", a cura di A.
Cusin e G. Leo

Writings by:J.
Altounian, S. Amati Sas, M. e M. Avakian, W. A.
Cusin, N. Janigro, G. Leo, B. E. Litowitz, S. Resnik, A.
Sabatini Scalmati, G. Schneider, M. �ebek,
F. Sironi, L. Tarantini.
Editore/Publisher: Edizioni Frenis Zero
Collana/Collection: Id-entit� mediterranee
Anno/Year: 2011
Pagine/Pages: 400
ISBN: 978-88-903710-4-2
Prezzo/Price: � 38,00
Click
here to order the book
"The Voyage Out" by Virginia
Woolf

Editore/Publisher: Edizioni Frenis Zero
ISBN: 978-88-97479-01-7
Anno/Year: 2011
Pages: 672
Prezzo/Price: � 25,00
Click
here to order the book
"Psicologia
dell'antisemitismo" di Imre Hermann

Author:Imre Hermann
Editore/Publisher: Edizioni Frenis Zero
ISBN: 978-88-903710-3-5
Anno/Year: 2011
Pages: 158
Prezzo/Price: � 18,00
Click
here to order the book
"Vite soffiate. I vinti della
psicoanalisi" di Giuseppe Leo

Editore/Publisher: Edizioni Frenis Zero
Edizione: 2a
ISBN: 978-88-903710-5-9
Anno/Year: 2011
Prezzo/Price: � 34,00
Click
here to order the book
"La Psicoanalisi e i suoi
confini" edited by Giuseppe Leo

Writings by: J.
Altounian, P. Fonagy, G.O. Gabbard, J.S. Grotstein, R.D.
Hinshelwood, J.P. Jim�nez, O.F. Kernberg, S. Resnik
Editore/Publisher: Astrolabio Ubaldini
ISBN: 978-88-340155-7-5
Anno/Year: 2009
Pages: 224
Prezzo/Price: � 20,00
"La Psicoanalisi. Intrecci Paesaggi
Confini"

Edited by S. Fizzarotti Selvaggi, G.Leo.
Writings by: Salomon Resnik, Mauro Mancia, Andreas Giannakoulas,
Mario Rossi Monti, Santa Fizzarotti Selvaggi, Giuseppe Leo.
Publisher: Schena Editore
ISBN 88-8229-567-2
Price: � 15,00
Click here to order the
book |
|
Introduction
Neuroimaging
approaches have been used to investigate neurobiological
changes in depressive patients in studies that address
remission after psychotherapy or antidepressant medication [1�2].
The majority of these studies focused on the effect of
treatment
on brain metabolism or perfusion at rest. A more recent series
of functional neuroimaging (fMRI) studies has turned to the use
of experimental tasks to examine processes that may be more directly
involved in emotional appraisal and control processes [3�6].
To date, studies examining the functional neuroanatomy of psychotherapy
in depressed patients have applied interpersonal therapy
or cognitive behavioural therapy [7�8]. The present research
reports on the first fMRI study with recurrently depressed patients
treated with psychodynamic psychotherapy. Long-term
psychodynamic
psychotherapy has been shown to be associated with
larger improvement in these difficult forms of depression than shorter
treatments [9�10].
The interest aroused
by neuroimaging studies of treatment of depression
is in part due to their potential importance in shedding light on the
mechanism of therapy. There is considerable evidence for
increased activation in limbic areas in depression, especially the amygdala,
under exposure to emotional stimuli [5�8,11�12]. This limbic
activation may be related to an increased reactivity of depressed
patients to emotional stimuli of negative valence [13]. It has
been proposed that antidepressant medication acts directly on this
abnormal reactivity [1], since amygdalar activation normalizes in
medicated remission [5�6]. However, changes associated with depression
have also been reported for prefrontal regions during the
execution of cognitive tasks [14]. Because of the regulatory nature
of many processes mapped onto the prefrontal cortex, these changes
may refer to mechanisms involved in emotional regulation during
the acute depressive episode, or reflect deficits in cognitive control
[15�16]. Different interpretations of signal changes in the prefrontal
cortex in depression or after therapy may be given [17].
Given
that emotion regulation may either have adverse or protective
roles in mental health, depending on the mechanism through
which it operates [18�19], it may be important to determine
not only if, but also how emotions are regulated [20].
Psychodynamic
approaches propose that depression, besides its biological
and social underpinnings, may be meaningfully conceived
as a specific organization of an individual�s conscious or
unconscious beliefs and feelings. The resulting mental operations
constitute defensive strategies that aim at avoiding negative
feelings arising out of conflict in order to maximize a subjective
sense of safety [21]. The objective of long-term psychodynamic
psychotherapy is a stable modification of these strategies
that allows the patient to work through, achieve insight, and
reappraise experiences that are related to depressive pathology.
Therefore, in our study we would expect neural patterns
of appraisal of sensitive material to change during therapy and
move from emotion regulation styles characterized by unfavourable
strategies to more integrated acceptance and awareness.
The identification of these neural patterns was the general
aim of our study.
To
date, only one fMRI study has investigated the effect of psychotherapy
(cognitive behavioural therapy) on major depression using
a standardized experimental task (Fu et al., [22]). In this study,
participants were exposed to faces portraying different degrees
of sadness. This study detected changes in the amygdalaanterior hippocampus
and posterior cingulate-precuneus regions as
well as the superior frontal gyrus, suggesting that areas
associated
in previous studies with increased reactivity to emotional
stimuli and mechanisms of control are also relevant for
psychotherapy effects.
In
the present study, patients with recurrent unipolar depression underwent
functional neuroimaging scans at the beginning of treatment
and after 15 months, during which they were treated with
psychodynamic psychotherapy by experienced therapists. A matched
healthy control group without psychotherapy was scanned
at the same time points. The stimulus materials were attachment-related
pictures from the Adult Attachment Projective Picture
System (AAP [23]), a measure that has been shown to be valid
for use in an fMRI environment [24�25]. These pictures are designed
to elicit mental engagement with attachment-related experiences
such as separation, illness, danger, and loss. The fundamental
ability to form attachment is indispensable for human social
relationships. Since Bowlby�s seminal contributions [26], attachment
and separation have become essential theoretical components
of developmental psychology, and psychodynamic theory
[27]. One key feature of interpersonal problems in depressed
patients is their feelings of helplessness and fear of losing
the love of a significant other [28]. Internal representations of
the self as unlovable and of attachment figures as unloving are a central
dimension of Beck�s cognitive triad of depression [29].
To
increase the capacity of the signal to elicit a response related to
the emotional processes of each individual, material was here prepared
using personalized content [11,30�32] derived from AAP
interviews with each participant. In the personally relevant
condition
the AAP attachment scenes were accompanied by individually
tailored descriptions containing core sentences from the
patient�s own narrative previously elicited by each picture (see Material
and methods for details). The same series of attachment scenes
accompanied by a standard factual, non-emotional description
for all participants was used as control condition.
In
addition to the detection of a neural signature of treatment, we
were interested to see the extent in which the pattern of change corresponded
or differed from the one described by Fu et al. [22].
On
the basis of this study, we formulated specific hypotheses relative
to the general aim of the study of characterizing changes in neural
patterns of appraisal and emotion regulation occurring after therapy.
Our first hypothesis was the normalization of emotional reactivity
indexed by changes in the amygdala-anterior hippocampus region,
as found by Fu et al. [22]. Our second hypothesis was
the existence of changes in prefrontal areas detected in previous
studies [22,31,33] as possible markers of the specific effect of
psychodynamic psychotherapy on styles of emotion regulation[1,8].
The
present design differed from that of previous studies in several
further respects. As mentioned before, neuroimaging studies
have examined the effects of short time psychotherapy (e.g.
12�20 weeks), applying cognitive-behavioural or interpersonal therapy
[7�8]. We examined depressed patients with a history of several
previous depressive episodes during psychodynamic treatment
providing a longer observation window (15 months of therapy)
than in previous studies.
The
present investigation therefore complements the existing emerging
picture of changes associated with the therapy of depression
emerging from neuroimaging studies. In meta-analyses of
neuroimaging studies of the effect of psychopharmacological intervention
[34], decreased activation following treatment was reported
in the anterior hippocampus and parahippocampal cortex,
in the subgenual and pregenual cingulus, in the insula, and the
putamen. In the prefrontal cortex, decreased activation was reported
in the middle and superior left frontal gyrus. The frontal lobes,
however, were also the seat of several meta-analytic foci of increased
activation following treatment (in the middle frontal gyri bilaterally
and the dorsal anterior cingulate cortex). Further increased
activation was found in the posterior cingulate cortex and
in temporo-parietal regions on both sides. With the exception of
Fu et al. [22], neuroimaging studies of psychotherapy of depression
have examined only resting state metabolism (for reviews,
see [7�8,35]). Here, therapy outcomes were associated primarily
with changes in the prefrontal cortex (dorsolateral, ventrolateral,
and medial) [36�38], but the direction of changes was
not entirely consistent. Some studies reported reduced rest metabolism
or perfusion that normalized at endpoint [37�38]. In other
studies, however, this finding was reversed [36]. When both psychotherapy
and pharmacotherapy were compared [37], there was
limited overlap between changes found in these two therapy modalities.
While all these studies appear to be broadly compatible with
the involvement of limbic and prefrontal areas, more studies of
task-related activation are needed to provide a comprehensive picture
of changes associated with remission.
Materials
and Methods
The
study protocol was approved by the ethical committee of the
University of Ulm and was in compliance with national legislation,
the principles expressed in the Declaration of Helsinki, and
the Code of Ethical Principles for Medical Research Involving Human
Subjects of the World Medical Association. All participants gave
written informed consent.
Participants
Patients
were recruited from the outpatient departments of two psychoanalytic
institutes in Bremen, Germany, and diagnosed by two
trained clinicians using the Structured Clinical Interview for DMS-IV
Diagnosis (SCID, German version, [39]). Eight patients
were
diagnosed with double depression (dysthymia and former major
depression episodes); in the remaining patients, degree of depression
was severe. Ten patients had a comorbid anxiety disorder.
Patients reported 3�10 previous depressive episodes (mean
6 episodes, standard deviation 3.4). Age at first occurrence of
depression was between 8 and 37 years (mean 20 years, standard
deviation 9.2). All patients reported previous unsuccessful psychopharmacological
and/or psychotherapeutic treatment (none
of which was psychodynamic). Exclusion criteria were other psychiatric
conditions as main diagnosis, substance abuse, significant
medical or neurological conditions (including medical causes
of depression) and psychotropic medication. Inclusion criteria
were a long history of recurrent major depression as an appropriate
indication for psychodynamic treatment. Nondepressed controls
were recruited from the community, matched for
age, sex and education; control participants had no history of previous
depressive episodes or other psychiatric conditions (SCID).
Ethical considerations precluded the recruitment of a depressed
group left untreated for the duration of the study. All participants
were right-handed.
Depression
severity and general psychological symptoms were assessed
using the Beck Depression Inventory (BDI, [40], German version
[41]) and the Global Severity Index (GSI) from the revised Symptom
Check List (SCL-90-R [42], German version [43]). Of
the
initial sample of 38 participants, 5 dropped out of the study (3 controls
and 2 patients). The final sample of 33 consisted of 16 patients
and 17 controls (7 males, mean age 38.9 years, standard devaition
12.4, range 20�64). Patients and controls in the final
sample
did not differ in age (logistic multiple regression, z
= 0.41, p
=0.68), sex (z
=0.49, p=
0.62), and education (z =
0.56, p=
0.57).
Treatment
Patients
were treated with long-term psychodynamic psychotherapy by
16 formally trained psychoanalysts (mean years of experience
22.4, standard deviation 7.9). Training as a psychodynamic psychotherapist
is certified by the German state and regulated
by laws specifying the hours of theory, psychotherapy under
supervision, and self-awareness training. Patients underwent 2
to 4 hours of therapy weekly (2 patients had 4 hours, 7 had 3
hours, and 7 had 2 hours per week). At the end of the study period
of 15 months, patients had received from 90 to 210 hours of
psychotherapy (mean 129 hours, standard deviation 37). Psychotherapy
formally qualified as psychodynamic on the basis of
two criteria. The first was observance of the ��couch setting��
in which
the patient talks while lying on a couch with no visual contact
with the therapist. The second were the core features of psychodynamic
thinking and therapeutic technique, which include interventions
focusing on the interpretation of the patient�s unconscious
conflicts as they emerge in the transference relationship
[21], and the focus on affect as it emerges in
relationships,
in attempts to avoid distressing thoughts, in memory of
past events, and in recurring patterns of interactions [44]. Adherence
to these principles was assured by a regular casediscussion group
led by one co-author (GB) in which all participating
therapists presented their patients and interventions. Participants
to this group had no access to the material produced by
patients during the AAP interview and its results used to create personalized
stimuli in the scanner.
All
patients were free of psychotropic medication throughout the
entire 15 months of the study by their own choice. After the end
of the study, therapy continued for varying periods of time, for total
therapy lengths ranging between 24 and 48 months in accordance
to individual therapeutic contracts, course of treatment, and
health insurance allowance.
Experimental
design
Stimuli
were derived from the Adult Attachment Projective Picture
System (AAP, [23]), an established and validated interview to
assess attachment patterns. The AAP consists of 7 picture stimuli,
designed to activate the attachment system [25]. Two to four
weeks prior to the fMRI experiment, one trained judge (ST) conducted
a standard AAP interview. Administration involves asking
participants in a semi-structured format to describe the scene
in the picture, including what characters are thinking or feeling,
and what they think might happen next. Three core sentences
that represented the attachment pattern of the participants
were extracted from the audiotaped responses to
each
AAP picture stimulus by two independent certified judges (e.g.,
��A girl is incarcerated in that big room��, ��My mother suffered
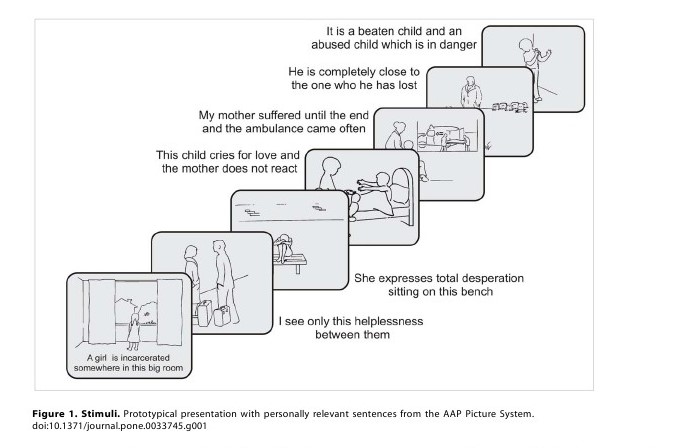
until the end and the ambulance came often��). These sentences
were paired to the respective picture to constitute the
��personally
relevant�� trials tailored to each participant (Figure 1).
The
same pictures, paired to sentences describing only the environment
of the depicted situation (e. g. ��There is a window with
curtains on the left and right��, ��There is a bed with a big blanket��)
constituted the ��neutral�� trials, and were identical for all
participants.
Each trial consisted of the presentation of the picturesentence pair
for 5 seconds, followed by a fixation cross between 7.5
and 12.5 seconds. Participants were instructed to mentally engage
with the attachment scene in the picture and its textual description.
Stimuli
were presented in two blocks of six trials each. Each block
contained three different sets of personally relevant sentences and
neutral sentences for each picture stimulus. Trials alternated between
the personally relevant and neutral in groups of seven AAP
picture stimuli. In total, there were 84 trials resulting in a scanning
time of about 21 min. After scanning, participants filled out
a questionnaire that asked to rate on a 7-point Likert scale the degree
of emotional arousal and relevance of the personally related sentences
(emotional arousal is here a construct referring to the degree
to which participants rated personal sentences emotionally involving,
not the degree of depressive symptoms associated with them).
There were no significant differences in the ratings of patients
and controls (t31 = 0.31, p= 0.38), thus suggesting that differences
in neural activation levels between patients and controls
were not driven by higher emotional valence of the stimuli.
Image acquisition
MRI
data were obtained with a 3-T Magnetom Allegra head scanner
(Siemens, Erlangen, Germany), equipped with a standard quadrature
head coil. To reduce anxiety levels, anatomical images were
acquired first (3D high resolution T1-weighted volume, MPRAGE-sequence;
TR/TE/TI = 2300/4.38/900 ms, flip angle =8u,
FOV= 25662566176 mm, isotropic voxel size 1 mm, total
acquisition time 7.5 min). A total of 508 EPI T2*-weighted whole
brain volumes were acquired (TR/TE = 2500/30 ms, flip
angle
90u, FOV 192 mm, matrix 64664, voxel size 363 mm, slice thickness
3 mm, 44 slices, interleaved acquisition order, standard AC-PC
orientation).
Statistical
analysis
Data
were analyzed and visualized using Brain Voyager QX1.10
(Brain Innovation, Maastricht, Netherlands). Volumes were
slice-time corrected and realigned to the first volume, normalized
into standard Talairach space with parameters obtained
from the co-registered high-resolution structural volumes, and
smoothed with a Gaussian isotropic kernel (8 mm full width-half
maximum). To remove low frequency drifts, data were high-pass
filtered (3 cycles, 3 sine waves within the extent of the data)
and z-transformed in each voxel separately. The BOLD response
function was modeled by convolving the trial onsets with a
standard hemodynamic response function. Motion-correction parameters
were included in the model as confounding covariates at
the first level. Effects of interest were estimated in each subject separately
and brought to the second level to account for a random effect
of subjects. Data were analyzed using a 2 (participant
group)62
(time)62 (sentence) factorial design. The main effect of interest
of the study was the interaction between the main effects of group
(patients vs. healthy control subjects) and time (baseline month
1 vs. endpoint month 15) on the activation detected by the
contrast
personally relevant vs. neutral. To identify regions associated
with changes we performed a whole-brain estimation of
the model voxel by voxel, considering a priori clusters larger than
20 voxels. Because effect sizes obtained in an interaction may be
expected to be small, we relied on the results of Fu et al. [22] to support
inference and considered the extent to which our result confirmed
findings of this previous study. The region of interest emerging
from this study, and from the comparison with the existing
literature on depression and its therapy, were the amygdala-anterior
hippocampus region, and prefrontal areas as markers
of emotion regulation processes [22,31,33], and more specifically
on the medial surface [22], given the inconsistency of reports
on the lateral surface/dorsolateral prefrontal cortex [45]. We
report on additional findings with explorative intent. Anatomical
identification of foci relied on publicly available
empirical
cytoarchitectonic maps [46�47] where these maps exist (amygdala
and hippocampus). In the regressions of clinical response
on signal changes, confounding by initial conditions was
avoided by including baseline scores (BDI or GSI) and signal
as
covariates in the model (as in ref. [31]).
Results
Clinical
Response
We
first looked at changes in depressive symptoms to ensure that
patients had responded to therapy. BDI scores refer to the level
of depressive symptoms, whereas GSI scores reflect general symptomatic
distress. At baseline, BDI score mean was 24.4 (standard
deviation 9.5, range 10 to 40). BDI scores from 10 to 18 suggest
mild to moderate depression; from 19 to 29 moderate to severe
depression [48]. At endpoint (i.e. at the time of the second scan),
the mean BDI score was 12.9 (standard deviation 8.2, range 2.5
to 35). Mean reduction in scores was 11.47 (paired t15
=4.99, p,0.001).
Mean GSI score at baseline was 1.35 (standard deviation
0.57, range 0.19 to 2.52), at endpoint 0.69 (standard
deviation
0.36, range 0.16 to 1.41). Mean GSI score reduction was 0.66
(paired t15
= 5.99, p,0.001).
Clinically, five patients still fulfilled
diagnostic criteria for major depressive disorder at endpoint.
All patients were planning at the end of the study to continue
psychotherapy.
At
baseline, controls showed scores in the healthy range in both GSI
(mean score 0.18, standard deviation 0.13) and BDI (mean score
2.17, standard deviation 2.48). GSI and BDI scores did not change
in controls. At endpoint, mean GSI score was 0.13 (standard
deviation 0.11) and mean BDI score was 1.94 (standard deviation
2.36; GSI: paired t16
=2.11, p=
0.052; BDI: paired t17
= 1.07, p=
0.299).
Neuroimaging
data
The
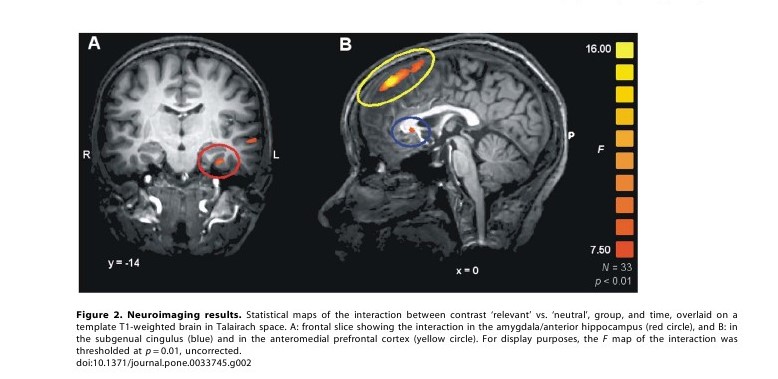
main effect of interest of the study was given by the interaction
between group (patients and controls) and time (pre vs. post)
for the contrast relevant vs. irrelevant, as this interaction directly
detects changes intervening during therapy that affected patients
but not controls in the appraisal of personalized material. In
this interaction an effect in the left amygdala was detected ([46], Talairach
coordinates x,
y, z:
233, 211,
224, F1,32
=9.00, p=
0.005), extending laterally into the anterior hippocampus where
it reached its peak in Brodmann area (BA) 36 (x,
y, z:
233, 214,
225, F1,32
=12.11, p =0.001,
Figure 2A, red circle), and towards
the middle temporal gyrus. On the right, the interaction failed
to reach significance (x,
y, z:
24, 210, 227,
F1,32
=3.24, p=
0.08), but this was consistent with the general left-lateralized pattern
of activation elicited by the task. Post-hoc analysis revealed this
effect to be due to patients showing more activity than control
subjects
at baseline (x,
y, z:
233, 214,
225, t32
= 2.81, p=
0.008), which
equalized or partially reversed at endpoint (t32
=22.41, p
=0.01).
An
interaction effect was also present in the ventral anterior cingulate
cortex (vACC, x,
y, z:
0, 23, 4, BA25, F1,32
= 6.91, p
=0.013, Figure 2B, blue circle).
Post-hoc analysis identified this interaction
as being mainly due to patients activating less when exposed
to self-referential material at endpoint than controls (t32
=22.1, p=
0.02), while at baseline the relation was partially inverted,
with patients activating more than controls (t32
= 1.74, p
=0.05). A fairly large area of
interaction involved the medial prefrontal
cortex in a much more dorsal position (x,
y, z:
3, 44, 49, BA8-9,
F1,32 =
13.47, p,0.001,
Figure 2B, yellow circle), which extended
onto the lateral aspect in the superior frontal gyrus and posteriorly
reached the middle frontal gyrus. This interaction was due
to cortical activation in patients at baseline relative to controls (t32
= 3.00, p=
0.003) that equalized at endpoint (t32
=21.6, n.s.).
No
other clusters of interaction in either direction were present with
a size larger than 150 mm3 (about 20 voxels), even when selected
at the liberal threshold p,0.05,
uncorrected.
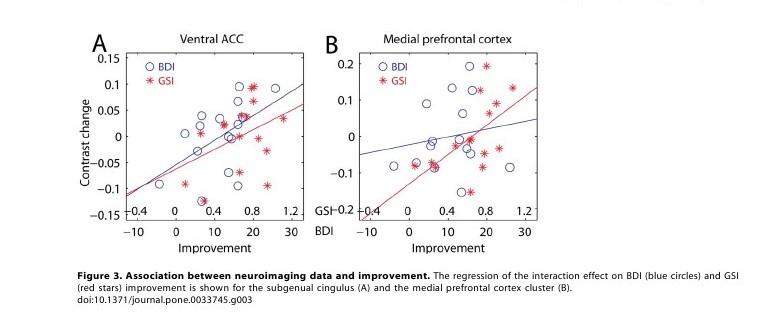
Correlation
of clinical response and cerebral activity
To
verify that the areas detected by the interaction were related with
changes in depressive symptoms, we regressed changes in the signal
of the contrast personally relevant vs. neutral on the improvement
in BDI scores, adjusting for initial levels to avoid
confounding
for initial severity. In the hippocampal/amygdalar cluster,
the association was only at trend level (t12
= 1.29, p=
0.11), accounting
for only 12% of the change in the signal from baseline to
endpoint. A stronger association was found in the ventral ACC
(t12
= 2.19, p=
0.02, explained variance 29%, Figure 3A, blue), which
did not change when age and sex were included as covariates
in the analysis (28%). In the medial prefrontal cortex the
association between the interaction effect and clinical response
was
not significant (t14
=0.92, p =0.19).
Long-term
psychodynamic psychotherapy is indicated for the treatment
of chronic depression [9], and may be expected to address
a wide range of issues that may complicate the depressive illness
and contribute to chronicity. For this reason, we extended our
analysis of the association of changes with response to the Global
Severity Index (GSI [42]), a more general index of psychic well
being than BDI. GSI and BDI scores were moderately associated
at baseline (r= 0.32, z = 1.80, p =0.04, 11% explained variance),
but strongly correlated at endpoint (r= 0.87, z =7.04, p,0.001,
76% of explained variance), suggesting effective treatment
of initial non-depressive symptoms captured by the GSI
score (interaction of the BDI/GSI association with time, t28
= 2.36, p= 0.01).
As
in the previous analysis with BDI scores, there was no significant
association between GSI improvement and the change in
the contrast signal in the hippocampal/amygdalar cluster (t12
= 1.08, n.s.). However, a significant association was found in the
ventral ACC cluster (t12 = 1.77, p= 0.05, explained variance 21%),
which was not changed by including sex and age as covariates
(20%). As one can see in Figure 3A, the association with GSI
improvement (in red) did not differ much from that of BDI (in blue).
Also in the medial prefrontal cortex there was an association between
the change in the contrast signal and clinical response measured
by GSI (t12 = 2.05, p= 0.03, explained variance 26%), which
remained after including age and sex as confounding covariates
(28%). As shown in Figure 3B, changes in the contrast signal
were more strongly associated with the general improvement measured
by GSI (in red) than with recovery from depression
as reflected by the BDI scores (in blue).
Discussion
When
exposed to personally attachment-relevant material, patients
undergoing long-term psychodynamic psychotherapy showed
changes in brain activation that were not observed in a sample
of control participants. The significant association of the changes
in the subgenual cingular and medial prefrontal cortex with
symptom improvement supported the hypothesis of their relevance
to the changes intervened during therapy.
Among
the areas involved by these changes, the anterior hippocampus/amygdalar
complex has been shown to be implicated in
the detection of emotional stimuli [49�52]
and displays
enhanced reactivity in depression [4�8,11�12,16] and anxiety [53�56].
The hippocampal/amygdalar correlate of change of the present
study fell within 5�10 mm of the anterior hippocampal area
associated with therapy change by Fu et al. [22] using cognitive
behavioral therapy. Changes in the ventral ACC were located
in the subgenual area reported by previous studies [36�37,57�59]
that provided converging evidence of its critical involvement
in both mood dysregulation and its resolution [60�61].
Changes over time in both areas consisted of a reduction in the
reactivity of these areas in response to personally relevant material
in patients. This pattern was not observed in controls.
A
third area, located anteriorly and superiorly in the medial prefrontal
cortex, was found in the present study to change from baseline
to endpoint in patients and not in controls. This area has been
associated with voluntary emotion regulation [19,62�63]. In
the
present study, this area displayed increased activation when exposed
at baseline to personally relevant material in patients, and equalized
at endpoint. Furthermore, changes in this area were associated
with changes in general symptom severity rather than with
changes in depressiveness specifically. This finding is consistent
with research showing this area�s general role in emotion
regulation and control processes.
It
is interesting that the prefrontal areas in which changes were detected
in the present study correspond to those associated with emotion
regulation [19,62�63]. Studies of emotional appraisal and regulation
have associated intentional avoidant and emotion
suppression
styles with increased psychiatric morbidity and vulnerability
to depression [19]. Because the material in the present
study was carefully chosen to represent personally relevant attachment
themes, we consider the associated emotional appraisal
to reflect the style of affect regulation.
There
were several aspects of this study that are worth commenting.
Among its strengths was the extension of neuroimaging approaches
to the investigation of changes during therapy in conditions
not investigated in previous studies: the recurrent type of
the depression from which patients suffered, the length of therapy,
and the psychodynamic nature of treatment. There were also
several limitations. First, while the personalized design may have
allowed an increase in validity and sensitivity of the stimuli used
in the scanner [11,30�32], it also introduces a possible confound
due to the existence of systematic differences in the material
produced by patients and controls. However, the absence of
differences in the rating of the emotional arousal of these personalized
sentences between patients and controls makes the existence
of this confound less likely. Second, the study lacked a natural
course control group composed by depressed patients on a waiting
list. Given the length of time during which the study was conducted,
keeping recurrent depressive patients on a waiting list would
have been unethical.
The
pattern of changes in prefrontal areas found in the present study
may be specifically associated with mechanisms of emotional appraisal
and control, suggesting reduced recourse to styles characterized
by suppression and avoidance after long-term therapy.
This interpretation outlines a possible mechanism for the
understanding of emotional appraisal and regulation in the psychodynamic
psychotherapy of depression. The relevance of these
finding for future studies rests in the possibility of documenting
specific mechanisms of action of depression therapy by
systematically collating results from different studies and comparing
different psychotherapeutic approaches, such as
psychodynamic,
behavioral, interpersonal, and psychopharmacological.
This
would be a first step towards monitoring progress of therapy
in individual patients.
|
|
References
1. DeRubeis RJ, Siegle GJ, Hollon SD (2008)
Cognitive therapy versus medication
for depression: Treatment outcomes and neural
mechanisms. Nature Rev
Neurosci 9: 788�796.
2. Ressler KJ, Mayberg HS (2007) Targeting
abnormal neural circuits in mood and
anxiety disorders: From the laboratory to the
clinic. Nat Neurosci 10:
1116�1124.
3. Buchsbaum MS, Wu J, Siegel BV, Hackett E,
Trenary M, et al. (1997) Effect of
sertraline on regional metabolic rate in
patients with affective disorder. Biol
Psychiatry 41: 15�22.
4. Davidson RJ, Irwin W, Anderle MJ, Kalin NH
(2003) The neural substrates of
affective processing in depressed patients
treated with venlafaxine.
Am J Psychiatry 160: 64�75.
5. Fu CHY, Williams SC, Cleare AJ, Brammer MJ,
Walsh ND, et al. (2004)
Attenuation of the neural response to sad faces
in major depression by
antidepressant treatment: A prospective,
event-related functional magnetic
resonance imaging study. Arch Gen Psychiatry 61:
877�889.
6. Sheline YI, Barch DM, Bonnelly JM, Ollinger
JM, Snyder AZ (2001) Increased
amygdala response to masked emotional faces in
depressed subjects resolves with
antidepressant treatment: An fMRI study. Biol
Psychiatry 50: 651�658.
7. Linden DEJ (2006) How psychotherapy changes
the brain. The contribution of
functional neuroimaging. Mol Psychiatry 11: 528�538.
8. Roffman JL, Marci CD, Glick DM, Dougherty DD,
Rauch SL (2005)
Neuroimaging and the functional neuroanatomy of
psychotherapy. Psych Med
35: 1385�1398.
9. Leichsenring F, Rabung S (2008) Effectiveness
of long-term psychodynamic
psychotherapy: A meta-analysis. JAMA 13: 1551�1565.
10. Leichsenring F, Rabung S (2011) Long-term
psychodynamic psychotherapy in
complex mental disorders: Update of a
meta-analysis. Br J Psychiatry 199:
15�22.
11. Siegle GJ, Steinhauer SR, Thase ME, Stenger
VA, Carter CS (2002) Can�t
shake that feeling: Event-related fMRI
assessment of sustained amygdala activity
in response to emotional information in
depressed individuals. Biol Psychiatry
51: 693�707.
12. Surguladze S, Brammer MJ, Keedwell P,
Giampietro V, Young AW, et al.
(2005) A differential pattern of neural response
toward sad versus happy facial
expressions in major depressive disorder. Biol
Psychiatry 57: 201�209.
13. Whalen PJ, Shin LM, Somerville LH, McLean
AA, Kim H (2002) Functional
neuroimaging studies of the amygdala in
depression. Seminars Clin Neuropsych
7: 234�242.
14. Thomas EJ, Elliott R (2009) Brain imaging
correlates of cognitive impairment in
depression. Front Hum Neurosci 3: 30. doi:10.3389/neuro.09.030.2009.
15. Bermpohl F, Walter M, Sajonz B, Lu�cke C,
Ha� gele C, et al. (2009) Attentional
modulation of emotional stimulus processing in
patients with major depression.
Alterations in prefrontal cortical regions.
Neurosci Lett 463: 108�113.
16. Erk S, Mikschl A, Stier S, Ciaramidaro A,
Gapp V, et al. (2010) Acute and
sustatined effects of cognitive emotion
regulation in major depression. J Neurosci
30: 15726�15734.
17. Taylor SF, Liberzon I (2007) Neural
correlates of emotion regulation in
psychopathology. Trends Cogn Sci 11: 413�418.
18. Fan Y, Wonneberger C, Enzi B, de Greck M,
Ulrich C, et al. (2011) The
narcissistic self and its psychological and
neural correlates: An exploratory fMRI
study. Psych Med 8: 1641�1650.
19. Ochsner KN, Gross JJ (2005) The cognitive
control of emotion. Trends Cogn
Sci 9: 242�249.
20. Phillips ML, Ladouceur CD, Drevets WC (2008)
A neural model of voluntary
and automatic emotion regulation: Implications
for understanding the
pathophysiology and neurodevelopment of bipolar
disorder. Mol Psychiatry
13: 829�857.
21. Fonagy P, Ka�chele H (2009) Psychoanalysis
and other long-term dynamic
psychotherapies. In: Gelder M, Andreasen N,
Lopez-Ibor J, Geddes J, eds. New
Oxford Textbook of Psychiatry. Oxford: Oxford
University Press, vol. 2. pp
1337�1350.
22. Fu CHY, Williams SCR, Cleare AJ, Scott J,
Mitterschiffthaler MT, et al. (2008)
Neural responses to sad facial expressions in
major depression following
cognitive behavioural therapy. Biol Psychiatry
64: 505�512.
23. George C, West M (2001) The development and
preliminary validation of a new
measure of adult attachment: The Adult
Attachment Projective. Attach Hum
Dev 3: 30�61.
24. Buchheim A, Erk S, George C, Ka�chele H,
Ruchsow M, et al. (2006) Measuring
attachment representation in an fMRI environment:
A pilot study. Psychopathology
39: 144�152.
25. Buchheim A, Erk S, George C, Ka�chele H,
Kircher T, et al. (2008) Neural
correlates of attachment trauma in borderline
personality disorder: A functional
magnetic resonance imaging study. Psych Res
Neuroimaging 163: 223�235.
26. Bowlby J (1982) Attachment and Loss. Vol 1:
Attachment. New York: Basic
Books (2nd ed.).
27. Levy KN (2005) The implications of
attachment theory and research for
understanding borderline personality disorder.
Dev Psychopathol 17: 959�986.
28. Taylor D (2008) Psychoanalytic and
psychodynamic therapies for depression:
The evidence base. Adv Psych Treatment 14: 401�413.
29. Beck AT, Rush AJ, Shaw BF, Emery G (1979)
Cognitive Therapy of Depression.
New York: Guildford Press.
30. Keedwell PA, Andrew C, Williams SCR, Brammer
MJ, Phillips ML (2005) The
neural correlates of anhedonia in major
depressive disorder. Biol Psychiatry 58:
843�853.
31. Siegle GJ, Carter CS, Thase ME (2006) Use of
fMRI to predict recovery from
unipolar depression with cognitive behaviour
therapy. Am J Psychiatry 163:
735�738.
32. Siegle G, Thompson W, Carter CS, Steinhauser
SR, Thase ME (2007)
Increased amygdala and decreased dorsolateral
prefrontal BOLD responses in
unipolar depression: Related and independent
features. Biol Psychiatry 61:
198�209.
33. Drevets WC, Price JL, Simpson JRJ, Todd RD,
Reich T, et al. (1997) Subgenual
prefrontal cortex abnormalities in mood
disorders. Nature 386: 824�827.
34. Fitzgerald PB, Laird AR, Maller J,
Daskalakis ZJ (2008) A meta-analytic study of
changes in brain activation in depression. Hum
Br Mapping 29: 683�695.
35. Frewen PA, Dozois DJA, Lanius RA (2007)
Neuroimaging studies of
psychological interventions for mood and anxiety
disorders: Empirical and
methodological review. Clin Psychol Rev 28: 228�246.
36. Brody AL, Saxena S, Stoessel P, Gillies LA,
Fairbanks LA, et al. (2001) Regional
brain metabolic changes in patients with major
depression treated with either
paroxetine or interpersonal therapy: Preliminary
findings. Arch Gen Psychiatry
58: 631�640.
37. Goldapple K, Segal Z, Garson C, Lau M,
Bieling P, et al. (2004) Modulation of
cortical-limbic pathways in major depression.
Arch Gen Psychiatry 61: 34�41.
38. Navarro V, Gasto� C, Lomena F, Mateos JJ,
Marcos T (2002) Normalization of
frontal cerebral perfusion in remitted elderly
major depression: A 12-month
follow-up SPECT study. NeuroImage 16: 781�787.
39. Wittchen HU, Wunderlich U, Gruschwitz S,
ZaudigM(1997) SKID-I Structured
Clinical Interview for DSM-IV (German Version).
Go�ttingen: Hogrefe.
40. Beck AT, Steer RA, Brown GK (1996) Beck
Depression Inventory. San Antonio
(TX): Psychological Corporation.
41. Hautzinger M, Bailer M, Worall H, Keller F
(1992) The Beck Depression
Inventory (German Version). Bern: Huber.
42. Derogatis LR (1993) BSI: Brief Symptom
Inventory. Minneapolis: National
Computer Systems.
43. Franke GH (1995) SCL-90-R (German Version).
Go�ttingen: Beltz.
44. Shedler J (2010) The efficacy of
psychodynamic psychotherapy. Am Psychol 65:
98�109.
45. Fitzgerald PB, Oxley TJ, Laird AR, Kulkarni
J, Egan GF (2006) An analysis of
functional neuroimaging studies of dorsolateral
prefrontal cortical activity in
depression. Psych Res Neuroimaging 148: 33�45.
46. Amunts K, Kedo O, Kindler M, Pieperhoff P,
Mohlberg H, et al. (2005)
Cytoarchitectonic mapping of the human amygdala,
hippocampal region and
entorhinal cortex: Intersubject variability and
probability maps. Anat Embryol
210: 343�352.
47. Eickhoff SB, Stephan KE, Mohlberg H, Grefkes
C, Fink GR, et al. (2005) A new
SPM toolbox for combining probabilistic
cytoarchitectonic maps and functional
imaging data. NeuroImage 25: 1325�1335.
48. Beck AT, Steer RA, Garbin MG (1988)
Psychometric properties of the Beck
Depression Inventory: Twenty-five years of
evaluation. Clin Psychol Rev 8: 77�100.
49. Davis M, Whalen PJ (2001) The amygdala:
Vigilance and emotion. Mol
Psychiatry 6: 13�34.
50. Morris JS, Frith CD, Perett DI, Rowland D,
Young AW, et al. (1996) A
differential neural response in the human
amygdala to fearful and happy facial
expressions. Nature 383: 812�815.
51. Phan KL, Wager T, Taylor SF, Liberzon I
(2002) Functional neuroanatomy of
emotion: A meta-analysis of emotion activation
studies in PET and fMRI.
NeuroImage 16: 331�348.
52. Phelps EA (2006) Emotion and cognition:
Insights from studies of the human
amygdala. Ann Rev Psychol 57: 27�53.
53. Gillath O, Bunge SA, Shaver PR, Wendelken C,
Mikulincer M (2005)
Attachment-style differences in the ability to
suppress negative thoughts:
Exploring the neural correlates. NeuroImage 28:
835�847.
54. Rauch SL, Whalen PJ, Shin LM, McInerney SC,
Macklin ML, et al. (2000)
Exaggerated amygdala response to masked facial
stimuli in posttraumatic stress
disorder: A functional MRI study. Biol
Psychiatry 47: 769�776.
55. Shin LM, McNally RJ, Kosslyn SM, Thompson WL,
Rauch SL, et al. (1999)
Regional cerebral bood flow during script-driven
imagery in childhood sexual
abuse-related PTSD: A PET investigation. Am J
Psychiatry 156: 575�584.
56. Beutel ME, Stark R, Pan H, Silbersweig D,
Dietrich S (2010) Changes of brain
activation pre- post short-term psychodynamic
inpatient psychotherapy: An
fMRI study of panic disorder patients. Psych Res
Neuroimaging 184: 96�104.
57. Drevets WC (2001) Neuroimaging and
neuropathological studies of depression:
Implications for the cognitive-emotional
features of mood disorders. Curr Opin
Neurobiol 11: 240�249.
58. Grimm S, Boesiger P, Beck J, Schuepbach D,
Bernpohl F, et al. (2009) Altered
negative BOLD responses in the default-mode
network during emotion
processing in depressed subjects.
Neuropsychopharmacology 34: 932�843.
59. Mayberg HS (2003) Positron emission
tomography imaging in depression: A
neural systems perspective. Neuroimaging Clin N
Am 13: 805�815.
60. Mayberg HS (2009) Targeted electrode-based
modulation of neural circuits for
depression. J Clin Invest 119: 717�725.
Neuroimaging and Psychotherapy of Depression
61. Carhart-Harris RL, Mayberg HS, Malizia AL,
Nutt D (2008) Mourning and
melancholia revisited: Correspondence between
principles of Freudian metapsychology
and empirical findings in neuropsychiatry. Ann
Gen Psychiatry 7: 9.
62. Ochsner KN, Ray RD, Cooper JC, Robertson ER,
Chopra S, et al. (2004) For
better or for worse: Neural systems supporting
the cognitive down- and upregulation
of negative emotion. NeuroImage 23: 483�499.
63. Goldin PR, Hutcherson CAC, Ochsner KN,
Glover GH, Gabrieli JDE (2005)
The neural bases of amusement and sadness: A
comparison of block contrast
and subject-specific emotion intensity
regression approaches. NeuroImage 27:
26�36.
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|



































