|
A.S.S.E.Psi.
web site (History of Psychiatry and Psychoanalytic Psychotherapy
)
A.S.S.E.Psi.NEWS
(to subscribe our monthly newsletter)
Ce.Psi.Di. (Centro
di Psicoterapia Dinamica "Mauro Mancia")
Maitres
� dispenser (Our reviews about psychoanalytic congresses)
Biblio
Reviews (Recensioni)
Congressi
ECM (in italian)
Events
(our congresses)
Tatiana Rosenthal
and ... other 'psycho-suiciders'
Thalassa.
Portolano of Psychoanalysis
PsychoWitz - Psychoanalysis and Humor (...per ridere un po'!)
Giuseppe Leo's Art
Gallery
Spazio
Rosenthal (femininity and psychoanalysis)
Psicoanalisi
Europea Video
Channel
A.S.S.E.Psi. Video
Channel
Ultima uscita/New issue:

"Fundamentalism
and Psychoanalysis"
Edited
by Giuseppe Leo
Prefaced
by: Vamik D. Volkan
Writings
by: L. Auestad W.
Bohleber S. Varvin L. West
Publisher:
Frenis Zero
Collection:
Mediterranean Id-entities
Year:
2017
Pages:
214
ISBN: 978-88-97479-13-0
Click
here to order the book

"Psicoanalisi, luoghi della resilienza
ed immigrazione"
Edited
by/a cura di:
Giuseppe Leo
Writings by/scritti di:
S.
Ara�jo Cabral, L.
Curone,
M. Francesconi,
L.
Frattini,
S.
Impagliazzo,
D. Centenaro Levandowski, G. Magnani, M. Manetti, C. Marangio,
G. A. Marra e Rosa, M. Martelli, M. R. Moro,
R. K. Papadopoulos, A. Pellicciari,
G. Rigon,
D.
Scotto di Fasano,
E. Zini, A. Zunino
Editore/Publisher: Edizioni Frenis Zero
Collection/Collana: Mediterranean
Id-entities
Anno/Year:
2017
Pagine/Pages:
372
ISBN:978-88-97479-11-6

"Psicoanalisi in Terra Santa"
Edited
by/a cura di: Ambra Cusin & Giuseppe Leo
Prefaced by/prefazione
di:
Anna Sabatini Scalmati
Writings by/scritti di:
H. Abramovitch A. Cusin M. Dwairy A. Lotem M.
Mansur M. P. Salatiello Afterword
by/ Postfazione
di:
Ch. U. Schminck-Gustavus
Notes by/ Note di: Nader Akkad
Editore/Publisher: Edizioni Frenis Zero
Collection/Collana: Mediterranean
Id-entities
Anno/Year:
2017
Pagine/Pages:
170
ISBN:978-88-97479-12-3

"Essere bambini a Gaza. Il trauma
infinito"
Authored
by/autore: Maria Patrizia Salatiello
Editore/Publisher: Edizioni Frenis Zero
Collection/Collana: Mediterranean
Id-entities
Anno/Year:
2016
Pagine/Pages:
242
ISBN:978-88-97479-08-6

Psychoanalysis,
Collective Traumas and Memory Places (English Edition)
Edited
by/a cura di: Giuseppe Leo Prefaced by/prefazione
di:
R.D.Hinshelwood
Writings by/scritti di: J. Altounian
W. Bohleber J. Deutsch
H. Halberstadt-Freud Y. Gampel
N. Janigro R.K. Papadopoulos
M. Ritter S. Varvin H.-J. Wirth
Editore/Publisher: Edizioni Frenis Zero
Collection/Collana: Mediterranean
Id-entities
Anno/Year:
2015
Pagine/Pages:
330
ISBN:978-88-97479-09-3

"L'uomo
dietro al lettino" di
Gabriele Cassullo
Prefaced
by/prefazione di: Jeremy
Holmes
Editore/Publisher: Edizioni Frenis Zero
Collection/Collana: Biografie
dell'Inconscio
Anno/Year:
2015
Pagine/Pages:
350
ISBN:978-88-97479-07-9
Prezzo/Price:
� 29,00
Click
here to order the book
(per Edizione
rilegata- Hardcover clicca qui)

"Neuroscience
and Psychoanalysis" (English Edition)
Edited by/a cura di: Giuseppe Leo Prefaced by/prefazione
di: Georg Northoff
Writings by/scritti di: D. Mann
A. N. Schore R. Stickgold
B.A. Van Der Kolk G. Vaslamatzis M.P. Walker
Editore/Publisher: Edizioni Frenis Zero
Collection/Collana: Psicoanalisi e neuroscienze
Anno/Year: 2014
Pagine/Pages: 300
ISBN:978-88-97479-06-2
Prezzo/Price: � 49,00
Click
here to order the book

Vera
Schmidt, "Scritti su psicoanalisi infantile ed
educazione"
Edited by/a cura di: Giuseppe Leo Prefaced by/prefazione
di: Alberto Angelini
Introduced by/introduzione di: Vlasta Polojaz
Afterword by/post-fazione di: Rita Corsa
Editore/Publisher: Edizioni Frenis Zero
Collana: Biografie dell'Inconscio
Anno/Year: 2014
Pagine/Pages: 248
ISBN:978-88-97479-05-5
Prezzo/Price: � 29,00
Click
here to order the book

Resnik,
S. et al. (a cura di Monica Ferri), "L'ascolto dei
sensi e dei luoghi nella relazione terapeutica"
Writings by:A.
Ambrosini, A. Bimbi, M. Ferri, G.
Gabbriellini, A. Luperini, S. Resnik,
S. Rodighiero, R. Tancredi, A. Taquini Resnik,
G. Trippi
Editore/Publisher: Edizioni Frenis Zero
Collana: Confini della Psicoanalisi
Anno/Year: 2013
Pagine/Pages: 156
ISBN:978-88-97479-04-8
Prezzo/Price: � 37,00
Click
here to order the book
Silvio
G. Cusin, "Sessualit� e conoscenza"

A cura di/Edited by: A. Cusin & G. Leo
Editore/Publisher: Edizioni Frenis Zero
Collana/Collection: Biografie dell'Inconscio
Anno/Year: 2013
Pagine/Pages: 476
ISBN: 978-88-97479-03-1
Prezzo/Price:
� 39,00
Click
here to order the book
AA.VV.,
"Psicoanalisi e luoghi della riabilitazione", a cura
di G. Leo e G. Riefolo (Editors)

A cura di/Edited by: G. Leo & G. Riefolo
Editore/Publisher: Edizioni Frenis Zero
Collana/Collection: Id-entit� mediterranee
Anno/Year: 2013
Pagine/Pages: 426
ISBN: 978-88-903710-9-7
Prezzo/Price:
� 39,00
Click
here to order the book
AA.VV.,
"Scrittura e memoria", a cura di R. Bolletti (Editor)

Writings by: J.
Altounian, S. Amati Sas, A. Arslan, R. Bolletti, P. De
Silvestris, M. Morello, A. Sabatini Scalmati.
Editore/Publisher: Edizioni Frenis Zero
Collana: Cordoglio e pregiudizio
Anno/Year: 2012
Pagine/Pages: 136
ISBN: 978-88-903710-7-3
Prezzo/Price: � 23,00
Click
here to order the book
AA.VV., "Lo
spazio velato. Femminile e discorso
psicoanalitico"
a cura di G. Leo e L. Montani (Editors)

Writings by: A.
Cusin, J. Kristeva, A. Loncan, S. Marino, B.
Massimilla, L. Montani, A. Nunziante Cesaro, S.
Parrello, M. Sommantico, G. Stanziano, L.
Tarantini, A. Zurolo.
Editore/Publisher: Edizioni Frenis Zero
Collana: Confini della psicoanalisi
Anno/Year: 2012
Pagine/Pages: 382
ISBN: 978-88-903710-6-6
Prezzo/Price: � 39,00
Click
here to order the book
AA.VV., Psychoanalysis
and its Borders, a cura di
G. Leo (Editor)

Writings by: J. Altounian, P.
Fonagy, G.O. Gabbard, J.S. Grotstein, R.D. Hinshelwood, J.P.
Jimenez, O.F. Kernberg, S. Resnik.
Editore/Publisher: Edizioni Frenis Zero
Collana/Collection: Borders of Psychoanalysis
Anno/Year: 2012
Pagine/Pages: 348
ISBN: 978-88-974790-2-4
Prezzo/Price: � 19,00
Click
here to order the book
AA.VV.,
"Psicoanalisi e luoghi della negazione", a cura di A.
Cusin e G. Leo

Writings by:J.
Altounian, S. Amati Sas, M. e M. Avakian, W. A.
Cusin, N. Janigro, G. Leo, B. E. Litowitz, S. Resnik, A.
Sabatini Scalmati, G. Schneider, M. �ebek,
F. Sironi, L. Tarantini.
Editore/Publisher: Edizioni Frenis Zero
Collana/Collection: Id-entit� mediterranee
Anno/Year: 2011
Pagine/Pages: 400
ISBN: 978-88-903710-4-2
Prezzo/Price: � 38,00
Click
here to order the book
"The Voyage Out" by Virginia
Woolf

Editore/Publisher: Edizioni Frenis Zero
ISBN: 978-88-97479-01-7
Anno/Year: 2011
Pages: 672
Prezzo/Price: � 25,00
Click
here to order the book
"Psicologia
dell'antisemitismo" di Imre Hermann

Author:Imre Hermann
Editore/Publisher: Edizioni Frenis Zero
ISBN: 978-88-903710-3-5
Anno/Year: 2011
Pages: 158
Prezzo/Price: � 18,00
Click
here to order the book
"Id-entit� mediterranee.
Psicoanalisi e luoghi della memoria" a cura di Giuseppe Leo
(editor)

Writings by: J.
Altounian, S. Amati Sas, M. Avakian, W. Bohleber, M. Breccia, A.
Coen, A. Cusin, G. Dana, J. Deutsch, S. Fizzarotti Selvaggi, Y.
Gampel, H. Halberstadt-Freud, N. Janigro, R. Ka�s, G. Leo, M.
Maisetti, F. Mazzei, M. Ritter, C. Trono, S. Varvin e H.-J. Wirth
Editore/Publisher: Edizioni Frenis Zero
ISBN: 978-88-903710-2-8
Anno/Year: 2010
Pages: 520
Prezzo/Price: � 41,00
Click
here to have a preview
Click
here to order the book
"Vite soffiate. I vinti della
psicoanalisi" di Giuseppe Leo

Editore/Publisher: Edizioni Frenis Zero
Edizione: 2a
ISBN: 978-88-903710-5-9
Anno/Year: 2011
Prezzo/Price: � 34,00
Click
here to order the book
"La Psicoanalisi e i suoi
confini" edited by Giuseppe Leo

Writings by: J.
Altounian, P. Fonagy, G.O. Gabbard, J.S. Grotstein, R.D.
Hinshelwood, J.P. Jim�nez, O.F. Kernberg, S. Resnik
Editore/Publisher: Astrolabio Ubaldini
ISBN: 978-88-340155-7-5
Anno/Year: 2009
Pages: 224
Prezzo/Price: � 20,00
"La Psicoanalisi. Intrecci Paesaggi
Confini"

Edited by S. Fizzarotti Selvaggi, G.Leo.
Writings by: Salomon Resnik, Mauro Mancia, Andreas Giannakoulas,
Mario Rossi Monti, Santa Fizzarotti Selvaggi, Giuseppe Leo.
Publisher: Schena Editore
ISBN 88-8229-567-2
Price: � 15,00
Click here to order the
book |
INTRODUCTION
The
observation of goal-directed actions done by another individual allows
the observer to achieve, typically, an immediate comprehension of what
that individual
is doing (see Rizzolatti et al., 2014). Besides goal, the observation
of a goal-directed action allows the observer to understand, on the
basis of how the
action is performed, the psychological state of the agent. It also
provides, in the case of interpersonal actions, an appraisal of the
affective/communicative qualities underlying the relation between the
agent and the action recipient. These aspects of action comprehension
have been named by Stern (1985, 2010) ��vitality affects�� or
��vitality forms��.
According to Stern (1985, 2010), the appraisal of vitality forms
depends on the kinematics properties of the observed movement (time,
space, force, direction). These movement properties create a
particular experience that reflects the affective/communicative state
of the agent. The capacity to express and understand the vitality
forms is already present in infants. These abilities denote a
primordial way to relate and to understand others and represent a
fundamental constitutive element of interpersonal relations (Stern,
1985, 2010; Trevarthen, 1998; Trevarthen and Aitken, 2001).
In a previous functional magnetic resonance imaging (fMRI) study (Di
Cesare et al., 2013) an attempt was done to define the brain areas
specifically involved in vitality form processing by comparing brain
activations during vitality forms judgment with respect to the
activations observed during goal understanding of the same action. The
results showed that a key structure involved in vitality forms
processing was the dorso-central sector of the insular cortex. These
data were confirmed by a further experiment in which participants had
to judge the vitality form of an action, imagine to perform it, and to
execute it (Di Cesare et al., 2015).
The aim of the present study was to assess using multi- voxel pattern
analysis (MVPA, Edelman et al., 1998; Haxby et al., 2001; Cox and
Savoy, 2003; Haynes and Rees, 2005; Norman et al., 2006; Kriegeskorte
et al., 2006; Kriegeskorte and Bandettini, 2007) whether observing an
action performed with different velocities will produce in the insula
distinct activation patterns according as to whether the participants
had to judge the action velocity or pay attention to its vitality form.
Videos showing actions performed with three velocities were selected
and presented to the participants. These velocities corresponded to
fast/rude (1.06 m/s), medium/neutral (0.57 m/s) and slow/gentle (0.38
m/s) velocities and vitality forms, respectively. These velocities
were selected on the basis of a preliminary behavioral experiment in
which participants observed actions performed with 12 different
velocities and had to judge them as very rude/very fast, rude/fast,
neutral/medium, gentle/slow, and very gentle/very slow, according to
the instructions.
The MVPA analysis showed the presence of a large number of
discriminative voxels with positive sign, that is exhibiting a
statistically significant preference for vitality, relative to
velocity while discriminative voxels exhibiting a statistically
significant preference for velocity were few. The insula sector
containing voxels with positive sign corresponded to the dorso-central
sector of the insula.
These findings indicate that the dorso-central insula does not encode
velocity parameters, but use this information to trigger the region
located in the dorso-central insula that previous data showed to be
involved in the control of the action style (Di Cesare et al., 2015).
These data provide strong support for the view that insula transforms
the physical aspects of an observed action in a communicative/affective
construct (vitality form). In virtue of this mechanism the observer is
able to understand the internal state of others.
MATERIALS
AND METHODS
Behavioral
Study
Subjects
Eighteen
healthy right-handed participants (mean age = 23.5 years, SD = 1.85
years) took part to the behavioral study. All participants had normal
or corrected-to-normal visual
acuity. They gave their written informed consent to the experimental
procedure, which was approved by the Local Ethics Committee (Parma,
Italy).
Stimuli
and Experimental Design
The
participants were shown video-clips representing two actors, one of
which moved an object (a bottle, a can, or a jar) with his right hand
towards the other actor. All three actions were performed with 12
different velocities (Figure 1). In all videos, the actor started from
the same initial position and reached the same final position. Figures
2A,B show the action performed with a jar. Each video lasted 2 s. A
total of 36 stimuli were presented (3 objects 12 velocities). The
experimental design was a 2 12 factorial with two levels of task (vitality,
velocity) and twelve levels of velocities (execution time from 500 ms
to 1600 ms).
Paradigm
and Task
The
experiment consisted of four experimental sessions. To avoid possible
influences of the velocity task on the vitality task, we presented the
vitality task before the velocity one. Thus, in the first two sessions,
participants were instructed to judge the vitality forms of the
actions, judge them as ��very rude��, ��rude��,
��neutral��, ��gentle��, or ��very gentle��
using a five point scale (vitality task). In the third and fourth
sessions, participants were asked to evaluate the velocity of the same
stimuli and to judge them as ��very fast��, ��fast��,
��medium��, ��slow��, and ��very slow�� using
again a five point scale (velocity task). Before the first and the
third experimental session, participants underwent a training session
(vitality training, before to start the session 1; velocity training,
before to start the session 3), with different stimuli from those used
during the experiment, to familiarize with the experimental procedures
and tasks.
Using
E-Prime Software, a total of 36 stimuli were presented for the
vitality and velocity tasks (3 actions, i.e., move a bottle, move a
jar, move a can, each one presented with 12 different velocity). Each
action was presented 10 times per task. Each experimental session
consisted of 180 trials presented in a randomized order. Each session
lasted about 10 min, the whole experiment lasting about 45 min.
The
velocity profile of each action was assessed by placing a reflective
marker on the object using 3D motion capture system (Vicon OMG, UK).
In particular, six infrared cameras (MX2 model) recorded the position
occupied by the marker in the 3D space for each action performed by
the actor with the object. After recording with Vicon Nexus at 100 Hz,
all recorded data were used to perform a kinematic analysis, using
MATLAB (The Mathworks, Natick, MA, USA) Software.
The
36 stimuli (3 objects 12 velocities) used in the experiment have been
compared by means of the Dynamic Time Warp (DTW; Berndt
and Clifford, 1994;
Ding et al., 2008)
metrics that allows to take into account the little differences in
duration of the trajectories. The DTW allows to measure the distance
between two time series that have different duration by finding the
correspondences between points in the time-series by

means
of a dynamic programming approach. This metrics has been applied to
the modulus of the velocity of each trajectory (and on vx, vy, vz
independently) and it produces a 36 36 matrix of pairwise distances.
The distance matrix had been analyzed for understanding if, for every
duration level, the distance among the objects inside each level of
velocity, is less than the ones of other duration levels. The results
of this analysis showed that there was no difference between the three
objects. For this reason we grouped the three objects and calculated
the velocity average profiles of the three objects (bottle, can, jar;
Figure 1).
fMRI
Studies
Participants
Sixteen
healthy right-handed volunteers [8 females (mean age = 24.1 years, SD
= 2 years, range = 21�28 years) and 8 males (mean age = 24.4 years,
SD = 2.18 years, range = 22�29 years)] participated in the
experiment. All participants had normal or corrected-to-normal visual
acuity. They gave their written informed consent to the experimental
procedure, which was approved by the Local Ethics Committee (Parma,
Italy).
Experimental
Design and Stimuli
The
experimental design was a 2 3 factorial with two levels of task (vitality,
velocity) and three levels of vitalities/velocities (gentle/slow,
neutral/medium, rude/fast). During the experiment, participants were
shown video-clips representing two male actors, one of which (the one
sitting on the left side of the screen) performed
an action towards the other actor using his right hand (Figures 2A,B).
To keep the observer�s attention, the action was executed using
three different objects (move a bottle, a can, a jar). All actions
were performed using three different velocities (execution times: 600,
1000, 1400 ms; mean velocity: 1.06, 0.57, 0.38 m/s; see Figure 2C).
These stimuli were selected on the basis of a previous behavioral
experiment. They mostly corresponded to fast/rude, medium/neutral and
slow/gentle velocity/vitality judgments (see also Supplementary
Figure 1). In all videos, the actor started from the same
initial position (Figures 2A,D) and reached the same final position (Figures
2B,D). Each video lasted 2 s. A total of nine stimuli were shown (3
objects 3 execution times).
Paradigm
and Task
Participants
lay in the scanner in a dimly lit environment. The stimuli were viewed
via digital visors (VisuaSTIM) with a 500,000 px 0.25 square inch
resolution and horizontal eye field of 30 . The digital transmission
of the signal to the scanner was via optic fiber. The software E-Prime
2 Professional (Psychology Software Tools, Inc., Pittsburgh, PA, USA, http://www.pstnet.com)
was used both for stimuli presentation and the recording of
participants� answers.
The
experiment was composed of four functional runs (2 for vitality task,
2 for velocity task). Similarly to the behavioral task, to avoid
possible biases elicited by the velocity condition on the vitality
form judgment, we presented the vitality form condition before the
velocity condition.

Thus,
in the first two runs, we presented
participants with video clips and asked them to pay attention to the
style of the action (vitality task). In the last two runs, we
presented participants with the same video clips and asked them to pay
attention to action velocity (velocity task). A fixation cross was
introduced in each video to restrain eye movements.
Every
run started with a white fixation cross, positioned at the center of a
black screen for 12 s. Each experimental trial presented a single
video-clip for 2 s followed by a jittered interval (fixation cross)
ranging 12�16 s. In 10% of cases, after 500 ms from video viewing,
the participants were cued presenting a task related question lasting
2.5 s. During this time, they had to provide an explicit response to
the stimuli (catch trials). More specifically, during the view of the
question cue (2.5 s), the participants had to indicate, on a response
box placed inside the scanner, whether the observed video was
rude/fast, neutral/medium, gentle/slow according to the task-type. In
total, participants viewed 50 video-clips (45 experimental trials, 5
catch trials) for each run, presented in a randomized order. Each
functional run lasted about 14 min.
Before
the first and the third experimental session, participants underwent a
training session (vitality training, before to start the session 1;
velocity training, before to start the session 3), with different
stimuli from those used during the experiment, to familiarize with the
experimental procedures and tasks.
fMRI
Data Acquisition
Anatomical
T1-weighted and functional T2 -weighted MR images were acquired with a
3 Tesla General Electrics scanner equipped
with an 8-channel receiver head-coil of the Department of Neuroscience
of University of Parma. Functional images were acquired using a T2
-weighted gradient-echo, echo-planar imaging (EPI) pulse sequence (acceleration
factor asset 2, 37 interleaved transverse slices covering the whole
brain, with a repetition time (TR) time of 2000 ms, echo time (TE) =
30 ms, flip-angle = 90 , field of view (FOV) = 205 205 mm2.
inter-slice gap = 0.5 mm, slice thickness = 3 mm, in-plane resolution
2.5 2.5 2.5 mm3).
Each scanning sequence comprised 416 interleaved volumes. Before the
third functional run, to allow participants to rest, a high-resolution
inversion recovery prepared T1-weighted anatomical scan was acquired
for each participant (acceleration factor arc 2, 156 sagittal slices,
matrix 256 256, isotropic resolution 1 1 1 mm3,
TI = 450 ms, TR = 8100 ms, TE = 3.2 ms, flip angle 12 ).
Statistical
Analysis
Univariate
Analysis
Data
analysis was performed with Brain Voyager QX (Brain Innovation). The
raw images were pre-processed in Brain Voyager QX performing the
following steps: sinc-interpolated slice-time correction, 3D motion
correction to correct small head movements, temporal high-pass
filtering to remove low frequency components up to seven cycles for
time course. Functional slices were then coregistered to the
anatomical volume and subsequently transformed into Talairach space.
All individual brains were segmented at gray/white matter boundary
using a semiautomatic procedure based on intensity values implemented
in Brain Voyager QX.
We applied
a minimal
amount of
spatial smoothing to
reduce the
residual effects
of head
movement (1-mm full-width
half-maximum (FWHM)
isotropic Gaussian
kernel).
TABLE
1 | Cerebral activity during (A) vitality forms vs. baseline; (B)
velocity vs. baseline.
|
|
|
|
|
Left
hemisphere
|
|
|
|
|
Right
hemisphere
|
|
|
|
Anatomical
region
|
|
|
|
|
|
|
|
|
|
|
|
|
|
x
|
y
|
z
|
t
|
|
x
|
y
|
z
|
t
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
(A)
Vitality forms vs. Baseline
|
10
|
26
|
|
|
|
|
|
|
|
|
|
Corpus
callosum
|
24
|
14.7
|
|
|
|
|
|
|
|
Medial
frontal gyrus
|
7
|
16
|
42
|
10.5
|
|
|
|
|
|
|
|
Middle
frontal gyrus
|
37
|
46
|
15
|
7.9
|
38
|
46
|
15
|
4.9
|
|
|
Supramarginal
gyrus
|
52
|
41
|
30
|
6.4
|
|
|
|
|
|
|
|
Superior
frontal gyrus
|
13
|
5
|
63
|
5.4
|
29
|
43
|
9
|
4.9
|
|
|
Middle
temporal gyrus
|
49
|
44
|
0
|
5.4
|
|
|
50
|
|
|
|
|
Precuneus
|
|
|
|
|
|
2
|
51
|
5.6
|
|
|
Inferior
frontal gyrus
|
|
|
|
|
|
50
|
37
|
3
|
5.3
|
|
|
Cerebellum
|
|
|
|
|
|
53
|
56
|
24
|
6.1
|
|
|
(B)
Velocity vs. Baseline
|
22
|
89
|
|
|
|
|
|
|
|
|
|
Middle
occipital gyrus
|
15
|
15.7
|
|
|
|
|
|
|
|
Cingulate
gyrus
|
10
|
13
|
42
|
9.8
|
|
|
53
|
|
|
|
|
Cerebellum
|
10
|
56
|
33
|
6.3
|
53
|
24
|
6.4
|
|
|
Middle
frontal gyrus
|
34
|
40
|
18
|
5.8
|
35
|
34
|
27
|
5.5
|
|
|
Middle
temporal gyrus
|
49
|
44
|
3
|
5.8
|
|
|
|
|
|
|
|
Precentral
gyrus
|
25
|
11
|
48
|
5.7
|
|
|
|
|
|
|
|
Inferior
frontal gyrus
|
49
|
7
|
30
|
5.6
|
|
|
53
|
|
|
|
|
Precuneus
|
|
|
|
|
|
5
|
42
|
6.5
|
|
|
Fusiform
gyrus
|
|
|
|
|
|
44
|
32
|
12
|
6.1
|
|
|
Post
central gyrus
|
|
|
|
|
|
35
|
20
|
30
|
6.0
|
|
|
Superior
frontal gyrus
|
|
|
|
|
|
23
|
55
|
12
|
5.7
|
|
|
Thalamus
|
|
|
|
|
|
17
|
11
|
15
|
5.6
|
|
 Local
maxima, as shown in � Supplementary Figure 2�, are given in Talairach brain
coordinates, significant threshold has been set at pFDR
< 0.05. Local
maxima, as shown in � Supplementary Figure 2�, are given in Talairach brain
coordinates, significant threshold has been set at pFDR
< 0.05.
Data
were analyzed using a random-effects model (
Friston et
al., 1999),
implemented in a two-level procedure. In the first level,
single-subject fMRI responses were modeled in a general linear model (GLM)
by a design-matrix comprising the onsets and durations of each event
according to the experimental task for each functional run.
In
the experiment, at the first level, for the task vitality we modeled
four regressors as follows: Rude, Neutral, Gentle, and Response; for
the task velocity we modeled other four regressors as follows: Fast,
Medium, Slow, and Response. The single video of each trial was modeled
as a mini epoch lasting 2 s. The Response for the first level analysis
was modeled with 2.5 s starting from the question was presented. In
the second level analysis (group-analysis), corresponding contrast
images of the first level for each participant were entered in a
random effects GLM (Friston et al., 2002).
This model was composed of six regressors (Fast, Medium, Slow, Rude,
Neutral, Gentle) and considered the pattern of activation obtained for
each level in the two tasks (vitality, velocity) vs. implicit baseline.
Within
this model, we assessed activations associated with each task vs.
implicit baseline (pFDR
< 0.05). This model did not reveal significant main effect of task
(vitality vs. velocity), levels (Rude vs. Gentle, Neutral vs. Gentle,
Rude vs. Neutral), or interaction.
The
location of the activation foci was determined in the Talairach
coordinates system. Those cerebral regions for which maps
are provided were also localized using the Talairach Client Software (version
2.4.3).
Testing
for Task-Complexity: Behavioral Analysis
Our
contrast of interest, vitality vs. velocity could have reflected some
effects associated with task presentation order such as a possible
fatigue effect. To test this possibility, we carried out a further
analysis, based on the responses given by the participants during the
scanning sessions when presented with the catch trials, i.e., those
trials in which the participants were required to give an explicit
response on the presented videos, indicating if they were rude,
neutral, gentle in terms of vitality form (vitality task) or fast,
medium, slow (velocity task). Ten responses were recorded for each
task for each participant. The dependent variable was the percent of
correct responses (��hits��). On these behavioral data, a GLM
analysis was carried out.
Multivoxel
Pattern Analysis in the Insula
A
multivoxel pattern analysis was then carried out to assess possible
different activation patterns in the insula in response to vitality
form (rude, neutral, gentle) and velocity (fast, medium, slow). We
decoded multivariate pattern of BOLD activation using support vector
machine (SVM) classifiers based on stimulus perception. On the basis
of our previous results (Di Cesare et al., 2013,
2015), we tested the activation pattern characterizing the
insular cortex in response to different action vitality forms (Rude,
Neutral, Gentle) compared to their velocities (Fast, Medium, Slow). We
built two regions of interest (ROIs), one at level of the left insula
(size of 1533 voxels) and one at the level of the right insula (size
of 1346 voxels). In order
to build the two ROIs, we drew a line between the border of the insula
and the parietal, frontal and temporal opercula cortices, which were
all excluded from the ROIs. To make sure that each drawn point
belonged to the insula, for each slice we checked the coordinates of 8
different border points with Talairach coordinates (Talairach Client�V.2.4.3).
We also built two control ROIs, one (CTRL 1) at level of the white
matter (size of 500 voxels, coordinates 28, 41, 26) and the other (CTRL
2) at level of Broadman Area 21 (BA 21) (size of 750 voxels,
coordinates 48, 4, 22). The control ROIs served to test results
reliability as a function of the multivoxel pattern model. All ROIs
were built on the mean anatomical structure of the participants. We
estimated the response of every voxel in each trial by fitting a
standard hemodynamic model to each voxel. The patterns of activation
related to each given trial consisted of the set of beta (% change)
values associated with one of the six predictors (task levels model)
for all voxels considered in the analysis. The Inter-Stimulus-Interval
ranged from 6 to 8 TRs (12�16 s). For each trial, one pre-onset
volume and 5 post-onset volumes were used to model the signal.
Since
the multivoxel pattern model required a comparison between tasks that
were presented in separate runs (vitality task: runs 1, 2; velocity
task: runs 3.4), we performed a cross-validation scheme considering
alternate runs (1.3; 2.4; 2.3; 1.4), dividing them in two different
groups (training runs and testing runs). More specifically, we trained
linear SVMs on the training datasets (e.g., from runs 1.3) and
evaluated the generalization of the model to new data (the test
datasets example e.g., from runs 2.4). This procedure was repeated for
four possible combinations (1.3 vs. 2.4; 2.4 vs. 1.3; 2.3 vs. 1.4; 1.4
vs. 2.3). To ascertain that this difference cannot be explained by
global effects such as amplitude differences between runs, we
conducted an additional ROI analysis considering only the voxels in
the left and right insula, testing for univariate differences between
vitality and velocity runs.
We
reported accuracies for the classification of new trials. Using
balanced datasets for training and testing (15 trials for each level,
rude/neutral/gentle; 15 trials for each level, fast/medium/slow), we
expected a rate higher than 50% (expected chance level, obtained with
1000 permutations, see Figure 4) for each different contrast (rude vs.
fast, neutral vs. medium, gentle vs. slow). The significance of this
difference was assessed by means of non-parametric Wilcoxon sign-rank
one-sided test (a = 0.05).
To
visualize the spatial activation patterns that were used for
classification and to assess consistency across participants, group
discriminative maps were created. For each participant, these maps
indicated the locations that contributed the most to the
discrimination of conditions. After using the linear SVM we ranked the
features (i.e., voxels) according to their contribution to the
discrimination at each individual map level and selected the peaks
through thresholding. For each participant, we selected the 50% most
discriminative voxels and created group discriminative maps
representing the overlap
between the
16 participants.
To calculate
a p-value for
each voxel, we used a Monte
Carlo simulation, where we randomly
selected 50% (or 35%) voxels from each subject, and
determined the overlap between subjects, under the assumption that the
spatial maps are completely unrelated. To account for the multiple
tests performed in creating these maps, we thesholded the maps using
false discovery rate (
Benjamin and Hochberg, 1995,
with q = 0.05), resulting in at least 10 of 16 participants. It is
worth noting that we obtained the same activation patterns selecting
35% threshold of most discriminative voxels with FDR corrected group
maps representing 8 of 16 participants. The classification accuracy
for each participant was always calculated with respect to the whole
set of features that did not depend on the threshold chosen for the
creation maps.
RESULTS
Behavioral
Study
The
participants� judgments obtained during vitality and velocity tasks
were automatically converted by E-Prime Software in numerical scores (very
rude/very fast = 5; rude/fast = 4; neutral/medium = 3; gentle/slow =
2; very gentle/very slow = 1). The scores were then modeled using a
GLM by a design matrix, comprising the participants� score related
to each task (vitality, velocity), for each execution time (12 levels).
The results of the GLM analysis indicate a significant difference in
judgments between the two Tasks (F.1,17/
= 10.07, p < 0.05, partial- 2
= 0.37, d=0.85). More specifically, the mean score for velocity task
(2.83; SD = 0.37) was shifted towards higher values relative to
vitality task (2.66; SD = 0.31), in spite of the fact that the stimuli
execution times were the same. In addition there was also a
significant difference in the judgments of the Execution Times (F.11,187/
= 310.37, p < 0.05, partial-2
= 1, d=1). The interaction Tasks Execution Times was also significant
(F.11,187/
= 5.54, p < 0.05, partial- 2
= 0.90, d = 0.89). Post hoc analysis revealed a significant difference
between Execution Times comparisons [1�2 (500�600 ms), 2�3
(600�700 ms), etc., p < 0.05 Newman Keuls corrected]. As shown in
Figure 3, for the interaction Task Execution Times, post hoc analysis
revealed a significant difference between vitality task and velocity
task in nine different comparisons (600, 700, 800, 900, 1000, 1100,
1300, 1400, 1500 ms; p < 0.05 Newman Keuls corrected).
The
analysis of the response times (RTs) revealed a difference between the
two Tasks (F.1,17/
= 13.8, p < 0.05, partial- 2
= 0.46, d = 0.93) showing that participants were significantly faster
in judging movement velocity (mean RT = 800 ms, SD = 220 ms) than
vitality forms (mean RT = 980 ms, SD = 295 ms). In addition there was
also a significant difference of RTs in the Execution Times (F.11,187/
= 4.3, p < 0.05, partial- 2
= 0.21, d = 1).
A
regression analysis was subsequently carried out to compare vitality
and velocity judgment (dependent variable) as a function of the
execution time (independent variable). As shown in Supplementary
Figure 1, the best fit curve representing the relation
between vitality perception and execution time follows a logarithmic
trend (R2
= 0.94, F = 3060. p < 0.00). The same relationship was also
observed for the velocity task (R2
= 0.87, F = 1513, p < 0.00). Taken
together, these data indicate that the fitting of the vitality and
velocity judgments as a function of the execution time, was very
similar.

fMRI Experiment
Response-Based Analysis Testing
This
analysis was based on the participants� responses (catch trials)
that were indicated in the scanner using a response box during
vitality and velocity tasks (see ��Materials and Methods��
Section). Within this analysis, we used the number of correct
responses (hits, i.e., subjects correct responses to specific velocity
or vitality, fast/rude�neutral/medium�gentle/slow) and RTs as
dependent variables to assess possible effects of the two task
difficulties. To this purpose, independent repeated measure GLM
analyses, with two levels of task (vitality and velocity) and three
levels of execution times (600, 1000, 1400 ms), were carried out. With
respect to hits, the results revealed no difference between tasks (p
> 0.05), showing that vitality and velocity were both judged
correctly. On the contrary, the analysis of RTs revealed a difference
between the two tasks (F.1,15/
= 7.7 p = 0.014, partial-!2
= 0.34, d = 0.74) showing that participants were significantly faster
in judging movement velocity (mean RT = 807 ms, SD = 116 ms) than
vitality forms (mean RT = 907 ms, SD = 102 ms). The dissociation
between accuracy and reaction time will be addressed in the discussion.
Univariate Analysis
Overall effect of ��vitality��
and ��velocity�� tasks
Observation
of the video-clips for each task (vitality and velocity) vs. implicit
baseline revealed a very similar activation pattern (
Supplementary Figure 2). In particular, there was a signal
increase in visual occipito-temporal areas, parietal lobe, SMA,
premotor and prefrontal cortex (for statistical values and coordinates
see Table 1). Additionally, insular activation was observed
bilaterally. The direct contrast vitality vs. velocity tasks and
velocity vs. vitality tasks, revealed no significant activations (p
> 0.05). Also the GLM analysis performed on the
insula did not reveal a significant effect of task (Left insula, t.15/
= 0.719, p = 0.48, Right insula, t.15/
= 0.618, p = 0.53).
Contrasts
between vitality forms levels and velocity levels
All
the direct contrasts within vitality task (Rude vs. Gentle, Rude vs.
Normal, Gentle vs. Normal, etc.,) and velocity task (Fast vs. Slow;
Fast vs. Medium; Slow vs. Medium, etc.,) did not reveal a significant
activation pattern.
Multivariate Pattern Analysis
The
multivoxel pattern analysis revealed that the classifier mean accuracy
for the levels across 16 participants was, for the left and right
insula, respectively: left 58.2% (Wilcoxon, one sided; p < 0.01)
and right 59.6% (p < 0.01) for the contrast rude vs. fast; left
58.8% (p < 0.01) and right 57.7% (p < 0.01) for the contrast
neutral vs. medium; left 56.7% (p < 0.01) and right 55.7% (p <
0.01) for gentle vs. slow (Figure 4). For the two control areas (CTRL
1, CRTL 2), the classifier mean accuracy across the same 16
participants was respectively: 51.5% (p > 0.05) and 51.6% (p >
0.05) for the contrast rude vs. fast; 51.9% (p > 0.05) and 51.8% (p
> 0.05) for the contrast neutral vs. medium; 50.9% (p > 0.05)
and 51.5% (p > 0.05) for gentle vs. slow, that is chance level
(Figure 4).
Subsequently,
group discriminative maps were constructed and inspected for
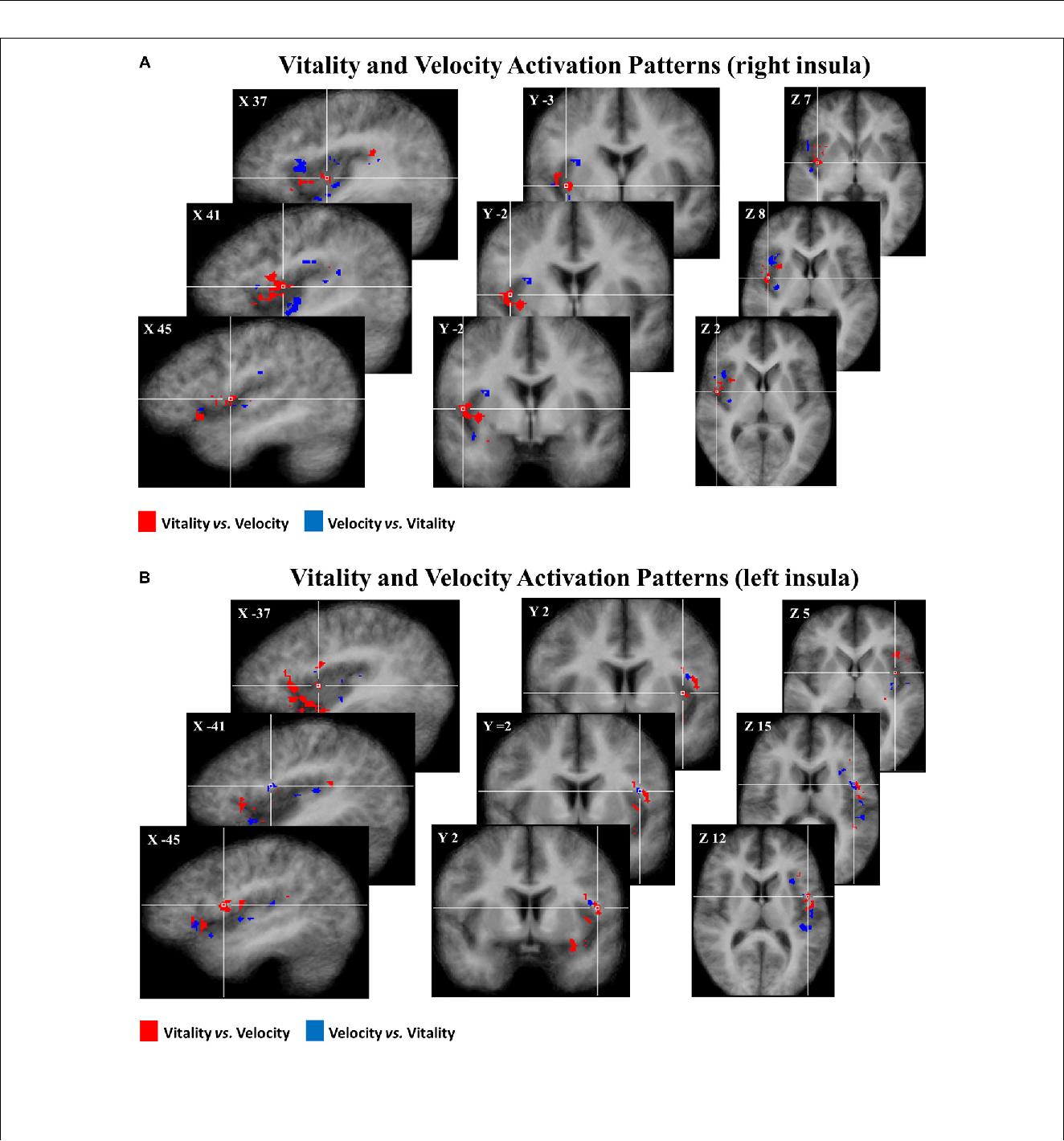
consistency of spatial activation patterns across participants. Figure
5 shows the pattern of discriminative voxels clustered in the insula.
The red color indicates positive weights, corresponding to voxels that
were more selective for vitality tasks with respect to velocity tasks,
while the blue color indicates negative weights corresponding to
voxels that were more selective for velocity tasks with respect to
vitality tasks. In the discriminative maps, the three different
comparisons (rude vs. fast, neutral vs. medium, gentle vs. slow) were
collapsed together.
In
addition, the multivoxel pattern analysis revealed that within each
task, the classifier mean accuracy for the comparisons among vitality
forms levels (i.e., rude vs. gentle, etc.) and velocity task (i.e.,
fast vs. slow, etc.) did not reach significance (p
> 0.05) (right insula: rude vs. gentle, 52%, fast vs. slow, 51.9%;
left insula: rude vs. fast, 51.8%, fast vs. slow, 50.7%).

DISCUSSION
In
his seminal book on mother-infant relationship, Stern
(1985) stressed that besides the goal and the intention of
the performing agent, there is a third, fundamental aspect that an
observer captures when viewing the actions of another individual: the
action vitality forms. Vitality forms characterize how an action is
performed and are detected on the basis of movement properties.
The
aim of the present study was to assess whether action velocity, one of
the crucial elements for understanding vitality forms, is encoded in
the insula as such, or velocity triggers the insula neural populations
encoding vitality forms. To this purpose we used multi-voxel pattern
analysis (MVPA) with the aim to establish whether in the insula there
are voxels discriminating vitality forms from velocity processing.
Before performing the fMRI experiment, we carried out a behavioral
study in which we presented arm actions performed at 12 different
velocities. The task of the participants was to judge either the
velocity or the vitality form of these actions. The results showed
that, although the stimuli presented in the two tasks were identical,
a significant difference was present in the subjects� judgment
according as to whether they were required to classify the observed
actions for their vitality form or their velocity. This should
indicate that the vitality form and velocity processing are two
different perceptual constructs. In accord with this conclusion are
also the reaction times results indicating that velocity processing
was significantly faster than vitality processing (mean velocity RT:
800 ms; mean vitality form RT: 980 ms).
The
neural bases of this finding are most likely due to the different
circuits that mediate the two tasks. A previous study (
Di Dio et al., 2013)
investigated the neural correlates of velocity processing during the
observation of actions performed by a biological effector (forelimb).
The results showed that the circuit included, beside visual-occipito
temporal areas and in particular MT/V5 and V6, a sector of the
superior parietal lobule, extending towards the intraparietal sulcus,
and the premotor cortex. As far as the insula is concerned there was
an activation of the rostralmost part of it, known to be involved in
cognitive tasks (
Kurth et al., 2010),
but not of the dorso-central part of the insula encoding vitality
forms. It is likely therefore that this cortical circuit, which was
found to be also activated in the present experiment, was responsible
for the fast RTs during the velocity task. In contrast, the necessity
to involve the dorso-central insula and to transform the velocity
information into vitality forms, required an additional time and was
therefore most likely responsible for longer RTs during vitality task.
On
the basis of the behavioral study, we also selected three actions,
corresponding to fast/rude (execution time: 600 ms; mean velocity:
1.06 m/s), medium/neutral (execution time: 1000 ms; mean velocity:
0.57 m/s) and slow/gentle (execution time: 1400 ms; mean velocity:
0.38 m/s) velocity/vitality judgments and used them for the fMRI study.
The
multivoxel pattern analysis revealed the presence of discriminative
voxels preferring vitality forms relative to velocity in the
dorso-central sector of the insula especially in the right hemisphere.
Our findings that the dorso-central part of the insula contains voxels
discriminating vitality forms are in agreement with recent findings on
the general functional organization of the insula in monkeys and
humans. More specifically, monkey experiments in which the insula
organization was studied by intracortical electrical stimulation
showed that the insula consists of different sectors endowed with
specific functional properties. The stimulation of the rostral sector
of insula determines positive ingestive behavior dorsally, and
negative ingestive behavior (e.g., disgust) ventrally (
Jezzini et al., 2012).
In contrast, the stimulation of the dorso-central sector of insula,
which most likely corresponds to the part activated in the present
experiment, elicits body parts movements with a rich representation of
the movements of the upper limb.
A
somehow similar organization pattern has been reported by Kurth
et al. (2010) in humans in a meta-analysis based on a very
large number of functional neuroimaging experiments. These authors
described four main distinct functional fields in the human insula:
the cognitive field, the sensorimotor, the olfactory-gustatory and the
socio-emotional. Except for the cognitive field that is not clear in
the monkey, there is a good correspondence in the two species between
the other fields. The sensorimotor field appears to correspond to the
sector involved in vitality form observation and production (Di Cesare
et al., 2013,
2015). In contrast, the rostral part of the insula
and its ventral part are related to classical Darwinian emotions (see
on this point Dolan,
2002; HYPERLINK \l
"page11" Phillips
et al., 2003;
Wicker et al., 2003;
Singer et al., 2004;
Pichon et al., 2009).
This functional characterization is in accord with the view of Stern
mentioned above that there is a fundamental difference between
vitality forms and the classical Darwinian emotions.
Some
very recent findings showed that the dorso-central insula is involved
in both vitality form execution and recognition suggesting
that neurons of this sector of the insula could be endowed with the
mirror mechanism (Di Cesare et al., 2015).
An interesting question concerns the output of the dorsal-central
insula and how this output may modulate the cortical circuits
underlying voluntary movements. A possible answer to this question may
come from some anatomical data obtained in the monkey. It has been
recently shown that the dorso-central sector of the insula has rich
connections with the parietal and frontal areas that form the circuit
involved in the organization of
arm
movements in the monkey (
Jeannerod et al., 1995;
HYPERLINK \l
"page11" Nelissen
and
Vanduffel, 2011)
and namely with areas AIP (
Borra et al., 2008),
F5 (Gerbella et al., 2011),
and 12r (Borra et al., 2011).
It is important to stress that a homologous parieto-frontal circuit
underlies arm movement organization also in humans (
Rizzolatti et al., 2014).
In
agreement with these findings, showing a connection between insula and
parieto-frontal circuit, are also the results of Almashaikhi
et al. (2014a,b) who stimulated electrically the middle and
posterior short gyri of the insula in patients with drug-resistant
epilepsy. The data showed that the stimulation of these insular
sectors determines evoked potential in the precentral gyrus and the
superior and inferior parietal lobules. These findings confirm the
connectivity of these sectors of the insula with the cortical areas
involved in the control of the voluntary movements as anatomically
demonstrated in the monkey.
In
conclusion, the main finding of our study is the demonstration that
the insula is a key area for vitality forms processing. During social
interactions, this area is triggered by the physical aspects of an
observed action determining in the observer a communicative/affective
construct (vitality form). In virtue of this mechanism, the observer
is able to understand the others� internal state. As shown recently
by Di
Cesare
et al.
(2015), this
mechanism is
also involved in
vitality form production (i.e., action execution) allowing an
individual to communicate his/her affective internal state to others.
|
| |
REFERENCES
Almashaikhi,
T., Rheims, S., Jung, J., Ostrowsky-Coste, K., Montavont, A., De
Bellescize, J., et al. (2014a). Functional connectivity of insular
efferences. Hum. Brain Mapp. 35, 5279�5294. doi: 10.1002/hbm.22549
Almashaikhi,
T., Rheims, S., Jung, J., Ostrowsky-Coste, K., Montavont, A., De
Bellescize, J., et al. (2014b). Intrainsular functional connectivity
in human. Hum. Brain Mapp. 35, 2779�2788. doi: 10.1002/hbm.22366
Benjamin,
Y., and Hochberg, Y. (1995). Controlling the false discovery rate: a
practical and powerful approach to multiple testing. J. R. Statist.
Soc. B 57, 289�300. doi: 10.2307/2346101
Berndt,
D. J., and Clifford, J. (1994). Using dynamic time warping to find
patterns in time series. In KDD workshop 10, 359�370.
Borra,
E., Gerbella, M., Rozzi, S., and Luppino, G. (2011). Anatomical
evidence for the involvement of the macaque ventrolateral prefrontal
area 12r in controlling goal-directed actions. J. Neurosci. 31,
12351�12363. doi: 10.1523/jneurosci. 1745-11.2011
Borra,
E., Belmalih, A., Calzavara, R., Gerbella, M., Murata, A., Rozzi, S.,
et al. (2008). Cortical connections of the macaque anterior
intraparietal (AIP) area. Cereb. Cortex 18, 1094�1111. doi: 10.1093/cercor/bhm146
Cox,
D. D., and Savoy, R. L. (2003). Functional magnetic resonance imaging
(fMRI) ��brain reading��: detecting and classifying
distributed patterns of fMRI activity in human visual cortex.
Neuroimage 19, 261�270. doi: 10.1016/s1053-8119(03)00049-1
Di
Cesare, G., Di Dio, C., Marchi, M., and Rizzolatti, G. (2015).
Expressing our internal states and understanding those of others. Proc.
Natl. Acad. Sci. U S A 112:10331�10335. doi: 10.1073/pnas.1512133112
Di
Cesare, G., Di Dio, C., Rochat, M. J., Sinigaglia, C.,
Bruschweiler-Stern, N., Stern, D. N., et al. (2013). The neural
correlates of ��vitality form�� recognition: an fMRI study.
Soc. Cogn. Affect. Neurosci. 9, 951�960. doi: 10.1093/scan/nst068
Di
Dio, C., Di Cesare, G., Higuchi, S., Roberts, N., Vogt, S., and
Rizzolatti, G. (2013). The neural correlates of velocity processing
during the observation of a
biological effector in the parietal and premotor cortex. Neuroimage
64, 425�436. doi: 10.1016/j.neuroimage.2012.09.026
Ding,
H., Trajcevski, G., Scheuermann, P., Wang, X., and Keogh, E. (2008).
��Querying and mining of time series data: experimental comparison
of representations and distance measures��, in Proceedings VLDB
Endowment 2, 1542�1552. Evanston, IL: Northwestern University
Dolan,
R. J. (2002). Emotion, cognition and behavior. Science 298,
1191�1194. doi: 10.1126/science.1076358
Edelman,
S., Grill-Spector, K., Kushnir, T., and Malach, R. (1998). Toward
direct visualization of the internal shape space by fMRI.
Psychobiology 26, 309�321.
Friston,
K. J., Glaser, D. E., Henson, R. N., Kiebel, S., Phillips, C., and
Ashburner, J. (2002). Classical and Bayesian inference in neuroimaging:
applications. Neuroimage 16, 484�512. doi: 10.1006/nimg.2002.1091
Friston,
K. J., Holmes, A. P., and Worsley, K. J. (1999). How many participants
constitute a study? Neuroimage 10, 1�5.
Gerbella,
M., Belmalih, A., Borra, E., Rozzi, S., and Luppino, G. (2011).
Connections of the anterior (F5a) subdivision of the macaque ventral
premotor area F5. Brain Struct. Funct. 216, 43�65. doi:
10.1007/s00429-010-0293-6
Haxby,
J. V., Gobbini, M. I., Fury, M., Ishai, A., Schouten, J. L., and
Pietrini, P. (2001). Distributed and overlapping representations of
faces and objects in ventral temporal cortex. Science 293,
2425�2430. doi: 10.1126/science.10 63736
Haynes,
J.-D., and Rees, G. (2005). Predicting the orientation of invisible
stimuli from activity in human primary visual cortex. Nat. Neurosci.
8, 686�691. doi: 10.1167/5.8.221
Jeannerod,
M., Arbib, M. A., Rizzolatti, G., and Sakata, H. (1995). Grasping
objects: the cortical mechanisms of visuomotor transformation. Trends
Neurosci. 18, 314�320. doi: 10.1016/0166-2236(95)93921-j
Jezzini,
A., Caruana, F., Stoianov, I., Gallese, V., and Rizzolatti, G. (2012).
The functional organization of the insula and of inner perisylvian
regions: an
intracortical microstimu-lation study. Proc. Natl. Acad. Sci. U S A
109, 10077�10082. doi: 10.1073/pnas.1200143109
Kriegeskorte, N., and Bandettini, P. (2007). Analyzing for information,
not activation, to exploit high-resolution fMRI. Neuroimage 38,
649�662. doi: 10. 1016/j.neuroimage.2007.02.022
Kriegeskorte, N., Goebel, R., and Bandettini, P. (2006).
Information-based functional brain mapping. Proc. Natl. Acad. Sci. U S
A 103, 3863�3868. doi: 10. 1073/pnas.0600244103
Kurth, F., Zilles, K., Fox, P. T., Laird, A. R., and Eickhoff, S. B.
(2010). A link between the systems: functional differentiation and
integration within the human insula revealed by meta-analysis. Brain
Struct. Funct. 214, 519�534. doi: 10.1007/s00429-010-0255-z
Nelissen, K., and Vanduffel, W. (2011). Grasping-related functional
magnetic resonance imaging brain responses in the macaque monkey. J.
Neurosci. 31, 8220�8229. doi: 10.1523/jneurosci.0623-11.2011
Norman, K. A., Polyn, S. M., Detre, G. J., and Haxby, J. V. (2006).
Beyond mind-reading: multi-voxel pattern analysis of fMRI data. Trends
Cogn. Sci. 10, 424�430. doi: 10.1016/j.tics.2006.07.005
Phillips, M. L., Drevets, W. C., Rauch, S. L., and Lane, R. (2003).
Neurobiology of emotion perception I: the neural basis of normal
emotion perception. Biol. Psychiatry 54, 504�514. doi:
10.1016/s0006-3223(03) 00168-9
Pichon, S., Gelder, B., and Grezes, J. (2009). Two different face of
threat. Comparing the neural systems for recognizing fear and anger in
dynamic body expressions. Neuroimage 1873�1883. doi:
10.1016/j.neuroimage.2009.03.084
Rizzolatti, G., Cattaneo, C., Fabbri-Destro, M., and Rozzi, S. (2014).
Cortical Mechanisms underlying the organization of goal-directed
actions and mirror neuron-based action understanding. Physiol. Rev.
94, 655�706. doi: 10. 1152/physrev.00009.2013
Singer,
T., Seymour, B., O�Doherty, J., Kaube, H., Dolan, R. J., Frith, C.
D. (2004). Empathy for pain involves the affective but not sensory
components of pain. Science 303, 1157�1162. doi:
10.1126/science.1093535
Stern,
D. N. (1985). The Interpersonal World of the Infant. (New York, NY:
Basic Books).
Stern,
D. N. (2010). Forms of Vitality Exploring Dynamic Experience in
Psychology, Arts, Psychotherapy, and Development. Oxford: Oxford
University press.
Trevarthen,
C. (1998). ��The concept and foundations of infant
intersubjectivity,�� in Intersubjective Communication and Emotion
in Early Ontogeny, ed. S. Braten (Cambridge: Cambridge University
Press).
Trevarthen,
C., and Aitken, K. J. (2001). Infant intersubjectivity: research,
theory and clinical applications. J. Child Psychol. Psychiatry 42,
3�48. doi: 10. 1017/s0021963001006552
Wicker
B., Keysers, C., Plailly, J., Royet, J. P., Gallese, V., and
Rizzolatti, G. (2003). Both of us disgusted in My insula: the common
neural basis of seeing and feeling disgust. Neuron 40, 655�664. doi:
10.1016/s0896-6273(03)00 679-2
|



























